|
Return
to Flumequine Index Page
Return
to Abstracts
Activity: Microbiocide
Structure:
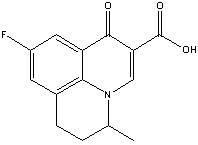
Adverse
Effects:
Bladder
Blood
Body
Weight Decrease
Bone
Eye
Liver
Mitogenic
Potent
Photosensitizer
Teratogenic
Contamination / Environmental
European
Commission: Not allowed to be used as an active ingredient
after July 25, 2003.
--
Flumequine's production and use as
an antibiotic(1) and antibiotic feed-additive on fish farms(2)
may result in its release to the environment through various
waste streams(SRC).
[(1) Budavari S, ed; The Merck Index. 13th ed. Whitehouse
Station, NJ: Merck and Co., Inc. p. 732 (2001) (2) Halling-Sorensen
B et al; Chemosphere 36: 357-93 (1998)]
-- Authorized or allowed for use in
aquaculture (2 day withdrawal time in France). ... Registered
or approved for use in aquatic or fishery situations in
Japan (20 mg/kg per day)
[Kirk-Othmer Encyclopedia of Chemical Technology. 4th ed.
Volumes 1: New York, NY. John Wiley and Sons, 1991-Present.,p.
V3 (1992) 618]
-- Ref:
Hazardous Substance Data Bank for Flumequine. Available
at Toxnet.
Effect
of cooking on residues of the quinolones oxolinic acid and
flumequine in fish.
Authors: STEFFENAK I HORMAZABAL V YNDESTAD M
Author Address: Dep. Food Hygiene, Norw. Coll. Vet. Med.,
P.O. Box 8146-Dep., N-0033 Oslo 1, Norway.
Source: ACTA VETERINARIA SCANDINAVICA; 35 (3). 1994.
299-301.
Abstract: BIOSIS COPYRIGHT:
BIOL ABS. The effect of cooking on residues of the quinolones
oxolinic acid and flumequine in fish was investigated. Salmon
containing residues of oxolinic acid and flumequine was
boiled or baked in the oven. Samples of raw and cooked muscle,
skin, and bone, as well as of the water in which the fish
was boiled and juice from the baked fish, were analysed.
Oxolinic acid and flumequine did not degrade at the temperatures
reached when cooking the fish. However,
fish muscle free from drug residues may be contaminated
during boiling and baking due to leakage of the drug from
reservoirs in the fish.
|
Bladder
(click
on for all fluorinated pesticides)
-- PubMed Abstract:
In order to elucidate the tumor-initiating potential of flumequine
(FL) in the liver, male C3H mice were given dietary administration
of 4000 ppm FL throughout the study or for 2 weeks at the initiation
stage, and then received 2 intraperitoneal injections of D-galactosamine
(Gal) at weeks 2 and 5, with or without 500 ppm phenobarbital
(PB) in their drinking water for 13 weeks to provide tumor-promoting
effects. Hepatocellular foci were observed in 2 out of 8 and 6
out of 7 animals in the FL/PB + Gal and FL/FL + Gal groups, respectively.
In addition, in an alkaline single-cell gel electrophoresis (comet)
assay that was performed using adult, infant, or partial hepatectomized
male ddY mice to evaluate the potential of FL at 500 mg/kg or
less, to act as a DNA damaging agent.
FL induced dose-dependent DNA damage in the stomach,
colon, and urinary bladder of adult mice at 3 h but
not at 24 h after its administration. Similarly, DNA damage was
noted in the regenerating liver and the livers of infant mice
at the 3 h time point. Furthermore, in in vitro assays that were
conducted to investigate the potential of FL to inhibit
eukaryotic topoisomerase II, which is responsible for the double-strand
DNA breakage reaction as well as bacterial gyrase, inhibitory
effects of FL on topoisomerase II were high relative to the influence
on bacterial gyrase. The results of our
studies thus strongly suggest that FL has initiating potential
in the livers of mice that is attributable to its induction of
DNA strand breaks.
Ref:
Toxicol Sci 2002 Oct;69(2):317-21; Mechanistic
study on flumequine hepatocarcinogenicity focusing on DNA damage
in mice; Y Kashida et al.
Blood
(click
on for all fluorinated pesticides)
BIOLOGICAL DATA 2.1. Hepatotoxicity and carcinogenicity. In short-term
and long-term studies of toxicity that were evaluated by the Committee
at its forty-second and forty-eighth meetings, oral administration
of flumequine caused dose-related hepatotoxic
effects in rats and CD-1 mice. The liver damage was most pronounced
in male mice, and included degenerative changes with hypertrophy,
fatty vacuolation, focal necrosis and increased mitotic activity.
After cessation of treatment with flumequine, the liver damage
was reversed. Treatment with flumequine had little or no effect
on P450-dependent hepatic drugmetabolizing enzymes or on glucuronyl
transferase. Flumequine increased the plasma
activities of alanine and aspartate aminotransferases, alkaline
phosphatase and lactate dehydrogenase. The overall no-observed-effect
level (NOEL) for hepatotoxic effects in mice was 25mg/kg bw per
day...
Ref: 2004
- Flumequine (addendum). First draft prepared by Mrs. M.E.J. Pronk,
Centre for Substances and Integrated Risk Assessment, National
Institute for Public Health and the Environment. Bilthoven, The
Netherlands.
http://www.fluorideaction.org/pesticides/flumequine.netherlands.2005pdf
Body
Weight Decrease
(click
on for all fluorinated pesticides)
-- the twice daily
oral administration of flumequine pellets 200 mg for 3 and 13
weeks at the dose level of 150 mg/kg bw/day induced few clinical
signs (vomiting, low food consumption),
marked reduction in bodyweight gain for females and minimal
to slight arthropathies with cartilage damage.
Only slight arthropathy was induced at the dose level of
60 mg/kg bw/day...
Ref: Committee for Veterinary Medicinal
Products. Flumequine. Summary Report. EMEA/MRL/104/96-FINAL. June
1996. Study performed by EMEA, London UK for the European Agency
for the Evaluation of Medicinal Products. Veterinary Medicines
Evaluation Unit.
http://www.fluoridealert.org/pesticides/Flumequine.Report.1996.pdf
Bone
(click on for all fluorinated pesticides)
-- Teratology studies
were conducted in rats (0, 100, 200 or 400 mg/kg bw), mice (50,
100, 200, and 400 mg/kg bw) and rabbits (100, 200 or 400 mg/kg
bw). None of these tests showed flumequine to be teratogenic or
embryotoxic, but at doses exceeding 100
mg/kg per day it does have an effect on bone formation.
The NOELs for the most sensitive species, rats and mice, were
100 mg/kg bw.
Ref: Committee for Veterinary Medicinal
Products. Flumequine. Summary Report. EMEA/MRL/104/96-FINAL. June
1996. Study performed by EMEA, London UK for the European Agency
for the Evaluation of Medicinal Products. Veterinary Medicines
Evaluation Unit.
http://www.fluoridealert.org/pesticides/Flumequine.Report.1996.pdf
-- the twice daily
oral administration of flumequine pellets 200 mg for 3 and 13
weeks at the dose level of 150 mg/kg bw/day induced few clinical
signs (vomiting, low food consumption),
marked reduction in bodyweight gain for females and minimal
to slight arthropathies with cartilage
damage. Only slight arthropathy was
induced at the dose level of 60 mg/kg bw/day...
Ref: Committee for Veterinary Medicinal
Products. Flumequine. Summary Report. EMEA/MRL/104/96-FINAL. June
1996. Study performed by EMEA, London UK for the European Agency
for the Evaluation of Medicinal Products. Veterinary Medicines
Evaluation Unit.
http://www.fluoridealert.org/pesticides/Flumequine.Report.1996.pdf
•
Arthropathy:
A rare form of chronic arthritis, reported to occur after attacks
of acute rheumatic fever, characterised by an unusual form of
bone erosion of the metacarpal heads and by ulnar deviation of
the fingers; it resembles rheumatoid arthritis, but with less
overt inflammation, and rheumatoid factor is absent. Synonym:
Jaccoud's arthropathy.
Defintion from: http://www.books.md/J/dic/Jaccoudsarthropathy.php
Eye
(click
on for all fluorinated pesticides)
--PubMed Abstract:
Flumequine (1 200 mg/day) was prescribed
as treatment for infection of the urinary tract to three patients
with chronic renal failure, who reported positive scotoma
three days later. Ophthalmologic examination
evinced bilateral symmetrical macular bullae. A characteristic
yellow papule was present at foveal level. In all three cases,
visual acuity was impaired (down to 4/10), without any angiographic
alteration. Foveolas showed a moderate persistent hyperfluorescence.
All patients recovered a normal visual acuity, within two days
after treatment cessation, and bullae disappeared without sequelae
within 5 days. The chronology and kinetics of clinical manifestations
were clearly and reproducibly correlated with flumequine therapy
in all patients, and suggest that this drug may be considered
responsible for the ocular symptom reported. Chronic renal
failure (creatinine clearance lower than 25 ml/mn) most certainly
favoured the appearance of visual troubles,
but other factors may possibly play a similar role: hepatic failure,
individual hypersensitivity... Quinolones used as urinary antiseptics
(nalidixic acid, oxolinic acid, pipemidic acid...), and other
flumequine analogues may possibly
be involved in such side-effects. This was reported by Bouissou
et al. in an experimental model with nalidixic acid, where transient
bullae appeared on young animals' articular cartilage. Such lesions
are related to focal alterations of the C2 intermediary layer
of cartilage, with marked edema of the interstitial material.
The volume of synovial fluid increases concomitantly. These
alterations suggest a direct cytotoxic effect at the intercellular
level of target organs, a mechanism possibly also occurring in
the retina.
Ref: J Fr Ophtalmol 1983;6(10):829-36. [Serous
macular detachment of the neuro-epithelium and flumequine].
[Article in French]. Sirbat D et al.
• Scotoma
definition: An island-like blind gap in the visual field. Taber's
Medical Dictionary
Liver
(click on for all fluorinated pesticides)
BIOLOGICAL DATA 2.1. Hepatotoxicity and carcinogenicity. In short-term
and long-term studies of toxicity that were evaluated by the Committee
at its forty-second and forty-eighth meetings, oral administration
of flumequine caused dose-related hepatotoxic
effects in rats and CD-1 mice. The liver damage was most pronounced
in male mice, and included degenerative changes with hypertrophy,
fatty vacuolation, focal necrosis and increased mitotic activity.
After cessation of treatment with flumequine, the liver damage
was reversed. Treatment with flumequine had little or no effect
on P450-dependent hepatic drugmetabolizing enzymes or on glucuronyl
transferase. Flumequine increased the plasma activities of alanine
and aspartate aminotransferases, alkaline phosphatase and lactate
dehydrogenase. The overall no-observed-effect level (NOEL) for
hepatotoxic effects in mice was 25mg/kg bw per day. The results
of long-term studies of toxicity that were evaluated by the Committee
at its forty-second meeting showed that flumequine had no carcinogenic
effects in rats, whereas in CD-1 mice an
increase in the incidence of liver tumours was observed at oral
doses of flumequine of ≥400mg/kg bw per day (the lowest
dose tested) in an 18-month study. The incidence of tumours in
male mice was significantly higher than that in female mice. In
male mice, the incidence of liver tumours increased in a dose-related
and time-dependent manner, and was paralleled by an increase in
the incidence of hepatotoxic changes. The present Committee
re-evaluated the three short-term studies in mice, which used
a two-stage hepatocarcinogenesis protocol, that were presented
to the Committee at its sixtieth meeting. In these studies, treatment
with flumequine caused the development of basophilic liver foci,
which could suggest that flumequine has tumour initiating potential.
However, the Committee also noted that concurrent hepatotoxicity
(evidenced by pale, vacuolated hepatocytes with fatty droplets,
inflammatory cell infiltration, increased mitotic figures and/or
necrosis) was observed, as well as a regenerative response to
these toxic changes and indications of oxidative stress.
Ref: 2004
- Flumequine (addendum). First draft prepared by Mrs. M.E.J. Pronk,
Centre for Substances and Integrated Risk Assessment, National
Institute for Public Health and the Environment. Bilthoven, The
Netherlands.
http://www.fluorideaction.org/pesticides/flumequine.netherlands.2005pdf
-- PubMed Abstract:
In order to elucidate the tumor-initiating potential of flumequine
(FL) in the liver, male C3H mice were given dietary administration
of 4000 ppm FL throughout the study or for 2 weeks at the initiation
stage, and then received 2 intraperitoneal injections of D-galactosamine
(Gal) at weeks 2 and 5, with or without 500 ppm phenobarbital
(PB) in their drinking water for 13 weeks to provide tumor-promoting
effects. Hepatocellular foci were observed in 2 out of 8 and 6
out of 7 animals in the FL/PB + Gal and FL/FL + Gal groups, respectively.
In addition, in an alkaline single-cell gel electrophoresis (comet)
assay that was performed using adult, infant, or partial hepatectomized
male ddY mice to evaluate the potential of FL at 500 mg/kg or
less, to act as a DNA damaging agent. FL
induced dose-dependent DNA damage in the stomach, colon, and urinary
bladder of adult mice at 3 h but not at 24 h after its administration.
Similarly, DNA damage was noted in
the regenerating liver and the livers of infant mice at the 3
h time point. Furthermore, in in vitro assays that were
conducted to investigate the potential of FL to inhibit eukaryotic
topoisomerase II, which is responsible for the double-strand DNA
breakage reaction as well as bacterial gyrase, inhibitory effects
of FL on topoisomerase II were high relative to the influence
on bacterial gyrase. The results of our
studies thus strongly suggest that FL has initiating potential
in the livers of mice that is attributable to its induction of
DNA strand breaks.
Ref:
Toxicol Sci 2002 Oct;69(2):317-21; Mechanistic
study on flumequine hepatocarcinogenicity focusing on DNA damage
in mice; Y Kashida et al.
-- 1999 PubMed Abstract:
It has been reported that flumequine
(FLU) induces
hepatic tumors in mice when given orally for 18 months.
We investigated possible underlying mechanisms using a two-stage
mouse hepatocarcinogenesis model. After initiation with a single
intraperitoneal injection of 100 mg/kg body weight diethylnitrosamine
(DEN) or saline, male CD-1 mice were given 4000 ppm FLU
in the diet or 500 ppm phenobarbital (PB) in drinking water for
9, 19, 24 or 30 weeks. Toxicity, evidenced
by centrilobular swollen and polar hepatocytes with fatty droplets,
infiltration of inflammatory cells and increased numbers of mitosis
in hepatocytes, was apparent in the livers of mice treated with
FLU at all time points, but its severity declined towards the
termination. FLU did not induce
cytochrome P-450 enzymes such as 1A1, 2B1 and 3A2 as assessed
immunohistochemically, while positive expression of 8-hydroxy-2'-deoxyguanosine
(8-OHdG) was increased in hepatocytes of both DEN + FLU
and FLU groups compared with the relevant controls. In
animals given PB, eosinophilic swelling of hepatocytes was prominent,
and the hepatocytes showed strongly positive reactions for CYP
1A1 and 3A2. Altered cell foci were induced in the livers of FLU-treated
animals both with and without DEN initiation, especially the former,
and their development paralleled the degree of hepatic toxicity.
These results suggest that FLU hepatocarcinogenicity
in mice is dependent on hepatotoxic damage and consequently increased
cell proliferation. Oxidative damage to DNA may also be
a crucial factor.
Ref: Cancer Lett 1999 Jul 1;141(1-2):99-107.
Hepatotoxicity
and consequently increased cell proliferation are associated with
flumequine hepatocarcinogenesis in mice. Yoshida M et al.
-- Maximum Residue
Limits. In calculating MRL values for flumequine, the following
factors were considered: á An ADI of 0-30 m g/kg, based on a toxicological
end-point, was established by JECFA. This will yield a daily intake
of 0-1800 m g/kg for a 60-kg person. á The parent drug was selected
as the marker residue. á Muscle
and kidney were proposed as target tissues. For
practical reasons, however, liver is the
proposed target tissue
for chickens in place of kidney.
Ref: Flumequine.
http://www.fao.org/docrep/W8338E/w8338e0a.htm#TopOfPage
-- In the 90-day subchronic
toxicity carried out on CD-1 mice, flumequine was administered
to at level doses of 0, 25, 50, 100, 400 and 800 mg/kg bw/day
for males and dosages of 1, 100, 400 and 800 mg/kg bw/day for
females. In the two high doses groups, the histopathological examination
of the livers revealed, in both males
and females, periacinar single cell necrosis
and inflammation, periacinar pigment laden
macrophages, increased ploidy of hepatocytes, hepatocytic intranuclear
inclusions, increased periacinar hepatocytic fatty vacuolation.
However, a periacinar hepatocytic hypertrophy was only observed
in males : in 7 of 12 animals of the 800 and 400 mg/kg bw dose
group, in 5 of 12 animals in the 100 mg/kg bw dose group and in
1 animal in the 50 mg/kg bw dose group and these lesions were
dosage-related. In addition, an inhibition of the activity of
NADPH-cytochrome P450 for females of the two highest dose grop
and of UDP-glucuronosyltranferase for males at 50 mg/kg bw was
also reported... 25 mg/kg bw/day was considered
as the NOEL for hepatotoxicity in mice.
-- In an 18-month carcinogeniciy study in mice, flumequine was
administered in the feed at 0, 400 or 800 mg/kg bw. The combined
incidence of benign and malignant liver
tumours was dose related : 37 % in the 400 mg/kg bw dose
group, 88 % in the high dose group vs. 9 % in the control group
for males and 13 % in the high dose females vs. 0 % for the contrl
and the low dose groups. Dose related changes in the hepatocytes
which paralleled the liver tumour incidence occurred in the low
dose males and in the high dose males and females.
-- There is evidence of compound-related tumorigenic
efffects in the liver of mice. In order to explain the
mechanism of liver tmour induction, the dosage of a preneoplastic
markter yGT and of a detoxification enzymes, GSH S-transferase,
were performed on liver samples collected in the 90-toxicity study
carried out in mice. No variations of yGT were noted whatever
the dosage used. However, an increase of the GSH S-transferase
activity in females dosed at 400 and 800 mg/kg bw and in males
dosed at 800 mg/kg bw showed that flllumequine induced detoxification
phenomena, showing cells hepatotoxicity. However, this phenomena
was not correlated with the number of tumours incidence. As the
tumorigenicity is considered to be a consequence of hepatotoxiciity,
it was concluded that the NOEL of 25 mg/kg bw/day covered both
end-points.
Ref: Committee for Veterinary Medicinal
Products. Flumequine. Summary Report. EMEA/MRL/104/96-FINAL. June
1996. Study performed by EMEA, London UK for the European Agency
for the Evaluation of Medicinal Products. Veterinary Medicines
Evaluation Unit.
http://www.fluoridealert.org/pesticides/Flumequine.Report.1996.pdf
Mitogenic
(click
on for all fluorinated pesticides)
PubMed Abstract: The
influence of flumequine on
mitogen induced lymphoid cell proliferation in European eels
(Anguilla anguilla L., 1758) was studied. For this purpose an
in vivo test, using peroral drug administration followed by successive
intraperitoneal injections with concanavalin A (ConA) or bacterial
lipopolysaccharides (LPS) and 5-bromo-2'-deoxyuridine, was applied.
Direct counting of proliferated cells in blood smears revealed
that flumequine possesses mitogenic properties.
A synergistic and an antagonistic effect of the drug was observed
after LPS and ConA stimulation, respectively. Flow cytometric
analysis of peripheral blood lymphoid cells showed a significant
reduction of the mean proportion surface immunoglobulin positive
cells in the flumequine-treated animals.
It is concluded that flumequine enhances
proliferation of lymphoid cells (probably surface immunoglobulin
negative cells) in eel under the present experimental conditions.
Ref: Vet Immunol
Immunopathol 1995 Jul;47(1-2):143-52. Influence
of flumequine on in vivo mitogen responses of European eel (Anguilla
anguilla L., 1758) lymphoid cells. van der Heijden MH et al.
Potent
Photosensitizer
(click
on for all fluorinated pesticides)
-- PubMed abstract:
An original physicochemical method is proposed for the evaluation
of the photosensitizing activity of drugs in vitro. A Nuclear
Magnetic Resonance (NMR) spectrum is recorded during light irradiation
of drug solutions. The change in the intensity of the NMR lines
under such conditions is termed the Photochemically Induced Dynamic
Nuclear Polarization (Photo-CIDNP) effect. It is related to the
formation of radical intermediates which may be involved in the
in vivo photosensitization reactions (the so-called type-I photoreactions).
Nine commercial quinolones were tested by this method: nalidixic,
oxolinic, pipemidic and piromidic acids, rosoxacin, flumequine,
enoxacin, pefloxacin and norfloxacin. Each quinolone was irradiated
in alcoholic solutions in its UV absorption band (300-350 nm)
in the absence or in the presence of a biological target chosen
as a model: the amino-acid N-acetyltyrosine. The quinolones were
classified in two groups in relation to the intensities of the
observed CIDNP effects. Nalidixic and oxolinic acids, rosoxacin
and flumequine are among the most potent
photosensitizers.
Ref: J Pharm Belg 1990 Sep-Oct;45(5):299-305.
[Photophysical
evaluation of photosensitization by various quinolones]. [Article
in French]. G Vermeersch et al.
• Definition:
Photosensitizer (sensitizer) is an agent that absorbs light
and subsequently initiates a photochemical or photophysical alteration
in the system, the agent being not consumed therewith. In case
of chemical alteration, the photosensitizer is usually identical
to a photocatalyst.
http://www.iupac.org/projects/posters01/parmon01.pdf
Teratogenic
(click on for all
fluorinated pesticides)
1999
PubMed Abstract:
(1) Follow-up studies of approximately 1,000 women exposed to
quinolones during pregnancy show no increase in the risk of malformations,
miscarriage, prematurity, intrauterine growth retardation or postnatal
disorders, but there are not enough data to draw firm conclusions.
(2) Teratogenic effects have been observed
in animals treated with the oldest quinolones (flumequine, nalidixic
acid and pipemidic acid) and also with sparfloxacin, a fluoroquinolone.
(3) Cartilage damage after postnatal exposure to quinolones in
animals and humans has been reported.
(4) Alternatives to quinolones can almost always be found for
pregnant women.
(5) Accidental exposure to quinolones during pregnancy does not
warrant termination.
Ref: Prescrire Int 1999 Feb;8(39):29-31.
Quinolones
and pregnancy: worrying animal findings, few clinical data.
[No authors
listed]
| Environmental
(click on for all fluorinated
pesticides)
--
Coyne et al. (1994) investigated the concentration of OTC
in the sediment of two cages at a fish farm site, and found
half-lives of 16 and 13 days. Oxytetracycline, oxolinic
acid, flumequine and sarafloxacine
were all found to be very persistent
in sediments (Hektonen et al. 1995). In the deeper
layer of the sediment hardly any degradation had occurred
after 180 days and a calculated half-life of more than 300
days was estimated. The residues in the top layer of the
sediment disappeared more rapidly. The removal of these
substances from the sediment is most probably due to leaching
and redistribution rather than degradation.
-- Samuelsen et al. (1994) showed that the toxicity of OTC
to bacteria declined rapidly in sediments, although no degradation
occurred. Binding to ions (Ca2+, Mg2+) and other substances
were mentioned as possible explanation for the inactivation
of oxytetracycline., The same study found that both oxolinic
acid and flumequine sustained their
antimicrobial activity over a six month period in sediment
material.
Ref: Environmental Project no. 659,
2002. Environmental Assessment of Veterinary Medicinal Products
in Denmark. 3. Environmental fate and occurrence of Veterinary
Medicinal Products. Danish Environmental Protection Agency.
http://www.mst.dk/udgiv/publications/2002/87-7944-971-9/html/kap03_eng.htm
Environmental
Spatio-temporal monitoring of the contamination of a coast
river in oxolinic acid, flumequine
and oxytetracycline
by
Raphael Delepee, Herve Pouliquen, Herve Le Bris
Unite
mixte de recherche INTRA/ENVN 1035 Chimiotherapie Aquacole
et Environment, Ecole Nationale Veterinaire de Nantes Atlanpole
- Le Chantrerie - BP 40706 Nantes Cedex 03, France
ABSTRACT:
Oxytetracycline, oxolinic acid and
flumequine are antibacterial agents commonly used
in fish farming, especially because of their broad spectrum
of activity. About 80 % of these drugs reached the environment
because of their administration as medicated pelleted feed
and their low oral bioavailability. Under these conditions,
there is a clear need in studying the impact of these treatments
on the freshwater environment.
A spatio-temporol
study was then realised to estimate the concentration of
oxolinic acid, flumequine and oxytetracycline in water,
sediments and bryophytes all along a coast river. The 25
sampling points were chosen around 6 study stations. Each
of these points were sampled once per season over one year.
Concentrations
in water were under limit of detection. The 900 analysis
showed that concentrations were greater in the bryophytes
• than in the sediments. The
greatest environmental concentrations were 120 ppb,
2000 ppb, 1500 ppb for oxolinic
acid flumequine and oxytetracycline
respectively. Multivariate statistical analysis were performed
on the data.
This
study showed a real contamination of the environment by
flumequine and oxytetracycline,
and to a lesser extent by oxolinic acid. No seasonal difference
in concentrations was noticed. The
analysis of the results showed the relevance of the use
of bryophytes instead of sediments in the freshwater environmental
monitoring. The fine study of the results seemed
to reveal a real impact of the study stations on the environment.
These observations should be confirmed by more specific
studies, by using moss bags for example.
Ref:
Abstract (Poster 7) from: Aquaculture and Environment Symposium,
September 18, 2002. 7th Bordeaux Aquaculture. September
18 - 20, 2002. Bordeau
•
Definition for bryophyte:
--
Any primitive plant in the division Bryophyta, includes
liverworts, mosses, and hornworts.
-- Plants in which the gametophyte generation is the larger,
persistent phase; they generally lack conducting tissues.
Bryophytes include the Hepaticophyta (liverworts), Anthocerotophyta
(hornworts), and Bryophyta (mosses). Ref: UCMP
Glossary: Botany
-- any plant of the phylum Bryophyta, having stems and leaves
but lacking true vascular tissue and roots and reproducing
by spores: includes the mosses and liverworts. [ETYMOLOGY:
19th Century: New Latin, from Greek bruon moss + -phyte]
bryophytic adjective. Ref: WordReference.com
|
|

