|
Return
to Isoxaflutole Index Page
Activity:
Herbicide
(cyclopropylisoxazole)
Structure:
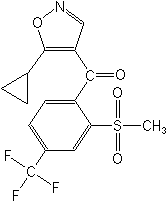
Adverse Effects:
Amyloidosis -
Systemic
Blood
Body
Weight Decrease
Bone
Cancer: Probable Human Carcinogen - LIVER,
THYROID
Cholesterol
Endocrine: Pituitary - disruption
in the thyroid-pituitary hormonal feedback mechanisms
Endocrine: Thyroid
Eye
Liver
Lymph node
Sciatic nerve
Environmental
As
of February 16, 2005, this herbicide is permitted in
or on 28 food commodities
in the United States - see list at bottom of page.
|
Amyloidosis
(click
on for all fluorinated pesticides)
An oncogenicity study
in mice administered 0, 25, 500 and 7,000 ppm with a NOEL of 25
ppm based on a slight effect on liver weight and body weight gain
at the LEL of 500 ppm. An increased incidence of hepatocellular
adenomas and carcinomas was observed at 7,000 ppm in both sexes.
Increased liver weight, non-neoplastic cellular changes in the
liver, and amyloidosis in the duodenum,
ileum, jejunum, kidneys, heart ventricle, mesenteric lymph node,
and thyroid were also observed at 7,000 ppm.
Ref: Federal Register: February 26, 1997
(Volume 62, Number 38)] [Notices] [Page 8737-8740]. Rhone-Poulenc
Ag Company; Pesticide Tolerance Petition Filing.
http://www.epa.gov/EPA-PEST/1997/February/Day-26/p4628.htm
Blood
(click
on for all fluorinated pesticides)
Short term toxicity. Target / critical
effect: Periacinar hypertrophy in liver, ocular lesions,
haematological effects. Lowest relevant
oral NOAEL / NOEL: 3 mg/kg bw/d, rat. Lowest relevant dermal NOAEL
/ NOEL: 100 mg/kg bw/d, 21d rat. Lowest relevant inhalation NOAEL
/ NOEL: no data available, not required.
April 2003 - Review report for the active
substance isoxaflutole. Isoxaflutole Sanco/3136/99-Final. 7 April
2003. Finalised in the Standing Committee on the Food Chain and
Animal Health at its meeting on 15 April 2003 in view of the inclusion
of isoxaflutole in Annex I of Directive 91/414/EEC.
http://www.fluorideaction.org/pesticides/isoxaflutole.eu.april.2003.pdf
Body
Weight Decrease
(click
on for all fluorinated pesticides)
-- In a chronic toxicity
study with dogs, a LOAEL was established 453 mg/kg/day for males
and 498 mg/kg/day for females, based on reduced
weight gains compared to controls and intravascular
hemolysis
with associated
clinical chemistry and histopathological findings. The NOEL is
44.81 mg/kg/day for males and 45.33 mg/kg/day for females.
-- In a combined chronic toxicity/carcinogenicity study in rats,
evidence of systemic toxicity was observed at 500 mg/kg/day and
included: abnormal gait, limited use of limbs, lower
body weight gains and food consumption,
decreased food efficiency during the first 14 weeks of
the study, elevated cholesterol levels throughout
the 104-week study, increased absolute and relative liver weights,
and thyroid hyperplasia. Increased incidence of periacinar hepatocytic
hypertrophy, portal tract (senile) bile duct changes, focal cystic
degeneration of the liver was observed in males at 20 mg/kg/day
and greater, females at 500 mg/kg/day. Eye opacity, gross necropsy
changes in eyes, corneal lesions, degeneration of sciatic nerve
and thigh muscles was observed in males at 20 mg/kg/day and higher
doses and in females at 500 mg/kg/day. The chronic LOAEL is 20
mg/kg/day based on liver, thyroid, ocular, and nervous system
toxicity in males and liver toxicity in females. The chronic
NOEL is 2.0 mg/kg/day.
-- In a developmental toxicity study in rats, maternal toxicity
was observed at 500 mg/kg/day, manifested as an increased incidence
of salivation, decreased body weight, weight
gain, and food consumption during the dosing period. The
maternal LOAEL is 500 mg/kg/day, based on increased incidence
of clinical signs and decreased body weights,
body weight gains, and food consumption. The maternal NOEL
is 100 mg/kg/day. Developmental toxicity, observed at 100 and
500 mg/kg/day, were manifested as increased incidences of fetuses/litters
with various anomalies: growth retardations
(decreased fetal body weight; increased
incidence of delayed ossification of sternebrae, metacarpals and
metatarsals).
In addition, an increased incidence of vertebral and rib anomalies
and high incidence of subcutaneous edema were observed at 500
mg/kg/day. The incidences of these anomalies were higher than
the concurrent control values and in some cases exceeded the range
for historical controls. The LOAEL for developmental toxicity
is 100 mg/kg/day, based on
decreased fetal body weights
and increased incidences of skeletal anomalies.
The developmental NOEL is 10 mg/kg/day.
-- In a developmental toxicity study in rabbits, maternal toxicity
was observed at 100 mg/kg/day, manifested as increased incidence
of clinical signs (little diet eaten and few feces) and
decreased body weight gain and food consumption during
the dosing period. The maternal LOAEL is 100 mg/kg/day, based
on increased incidence of clinical signs, decreased
body weight gains and food consumption. The maternal NOEL
is 20 mg/kg/day. Developmental toxicity, observed at 5 mg/kg/day,
consisted of increased incidence of 27th
pre-sacral vertebrae. Additional findings noted at 20 and
100 mg/kg/day were manifested as increased number of postimplantation
loss and late resorptions, as well as growth
retardations in the form of generalized reduction in skeletal
ossification, and increased incidence of 13 pairs of ribs. At
100 mg/kg/day, an increased incidence of fetuses with incisors
not erupted was also observed. Incidences of these anomalies,
on a litter basis, were higher than the concurrent control values
and in some cases exceeded the range for historical controls.
The LOAEL for developmental toxicity is 5 mg/kg/day, based on
increased incidence of fetuses with 27th pre-sacral vertebrae.
The developmental NOEL was not established.
-- Reproductive Toxicity. In a 2-generation reproduction study
in rats, evidence of toxicity was observed in the male and female
parental rats of both generations: at 20 and 500 mg/kg/day, increased
absolute and relative liver weights associated
with liver hypertrophy was observed; at 500 mg/kg/day (HDT),
decreased body weight, body weight gain
and food consumption during premating and gestation, and
increased incidence of subacute inflammation of the
cornea of the eye
in F0 adults as well as keratitis in F1 adults were reported.
There were no other systemic effects that were attributed to treatment,
nor was there any indication, at any treatment level, of an effect
on reproductive performance of the
adults. Treatment-related effects were observed in F1 and F2 offspring:
at 20 and 500 mg/kg/day, reduction in pup survival was noted;
at 500 mg/kg/day, decrease in body weights
of F1 and F2 pups throughout lactation, increased incidence of
chronic keratitis, low incidence of inflammation of the iris,
as well as retinal and vitreous bleeding in F2 pups and weanlings
were observed. Necropsy of F1 and F2 pups culled on Day 4 revealed
an increased number of pups with no milk in the stomach and underdeveloped
renal papillae. The Systemic LOAEL is 17.4 mg/kg/day for males
and females, based upon increased liver weights and hypertrophy
and the Systemic NOEL is 1.76 mg/kg/day for males and females.
The Reproductive LOAEL is greater than 437 mg/kg/day, based on
lack of reproductive effects and the Reproductive NOEL is greater
than or equal to 437 mg/kg/day.
-- In a subchronic neurotoxicity study in rats, treatment-related
effects observed in high-dose males consisted of decreases
in body weight and body weight gain. The LOAEL was established
at 25 mg/kg/day based on significant decreases in mean hind limb
grip strength in male rats at 25 mg/kg/day (LDT) during both trials
at week 13 as well as a non-significant decrease in mean forelimb
grip strength at week 13.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Bone
(click
on for all fluorinated pesticides)
-- In a developmental
toxicity study in rats, maternal toxicity was observed at 500
mg/kg/day, manifested as an increased incidence of salivation,
decreased body weight, weight gain, and food consumption during
the dosing period. The maternal LOAEL is 500 mg/kg/day, based
on increased incidence of clinical signs and decreased body weights,
body weight gains, and food consumption. The maternal NOEL is
100 mg/kg/day. Developmental toxicity, observed at 100 and 500
mg/kg/day, were manifested as increased incidences of fetuses/litters
with various anomalies: growth
retardations (decreased fetal body weight;
increased incidence of delayed ossification of sternebrae, metacarpals
and metatarsals).
In addition, an increased incidence of vertebral
and rib anomalies and high incidence of
subcutaneous edema were observed at 500 mg/kg/day.
The incidences
of these anomalies were higher than the
concurrent control values and in some cases exceeded the range
for historical controls. The LOAEL for developmental toxicity
is 100 mg/kg/day, based on decreased fetal
body weights and increased incidences
of skeletal anomalies. The developmental NOEL is 10 mg/kg/day.
-- In a developmental toxicity study in rabbits, maternal toxicity
was observed at 100 mg/kg/day, manifested as increased incidence
of clinical signs (little diet eaten and few feces) and
decreased body weight gain and food consumption during the dosing
period. The maternal LOAEL is 100 mg/kg/day, based on increased
incidence of clinical signs, decreased body weight gains and food
consumption. The maternal NOEL is 20 mg/kg/day. Developmental
toxicity, observed at 5 mg/kg/day, consisted of increased
incidence of 27th pre-sacral vertebrae. Additional findings
noted at 20 and 100 mg/kg/day were manifested as increased number
of postimplantation loss and late resorptions, as well as growth
retardations in the form of generalized reduction in
skeletal ossification, and increased incidence of 13 pairs of
ribs. At 100
mg/kg/day, an increased incidence of fetuses
with incisors not erupted was also observed.
Incidences of these anomalies, on a litter basis, were higher
than the concurrent control values and in some cases exceeded
the range for historical controls. The LOAEL for developmental
toxicity is 5 mg/kg/day, based on increased
incidence of fetuses with 27th pre-sacral vertebrae. The
developmental NOEL was not established.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Cancer:
Probable Human Carcinogen - LIVER, THYROID
(click
on for all fluorinated pesticides)
Likely
to be carcinogenic to Humans. Statistically
significant increases in liver tumors
in both sexes of CD-1 mice & Sprague-Dawley rats; statistically
significant increases in thyroid
tumors in male rats.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Likely
to be carcinogenic to humans.
Reviewed 8/ 6/ 97.
Ref:
List of Chemicals Evaluated for Carcinogenic Potential. Science
Information Management Branch, Health Effects Division, Office
of Pesticide Programs, U. S. Environmental Protection Agency.
March 15, 2002.
http://www.biomuncie.org/chemicals_evaluated_for_carcinog.htm
-- Isoxaflutole demonstrates developmental toxicity and has been
classified as a Group B2 carcinogen (probable
human carcinogen).
-- In a combined chronic toxicity/carcinogenicity study in rats,
evidence of systemic toxicity was observed at 500 mg/kg/day...
Under the conditions of this study, isoxaflutole induced benign
and malignant tumors of the liver in both sexes at 500 mg/kg/day
hepatocellular adenomas and hepatocellular carcinomas.
Combined incidences of liver adenoma/carcinoma in males and females
showed animals bearing carcinomas in the
majority. Thyroid follicular adenomas
occurred with increased frequency in 500 mg/kg/day males.
The tumor incidences exceeded the historical
incidence of these tumors for this strain in the laboratory.
The study demonstrated that isoxaflutole is carcinogenic to rats
at a dose of 500 mg/kg/day. The chemical was administered at a
dose sufficient to test its carcinogenic potential. At 500 mg/kg/day,
there were alterations in most of the parameters measured including
clinical signs of toxicity, body weight gain, food consumption,
food conversion efficiency, and clinical as well as post-mortem
pathology. Thyroid stimulating hormone (TSH) was not measured
in this study. However, in a separate special study investigating
the mechanism of action of isoxaflutole on the thyroid, tested
at the same doses as this study, TSH was indirectly measured since
there was a significant reduction in T4
level and thyroid gland weights were significantly increased.
These results were sufficient to support the hypothesis that isoxaflutole
may have induced thyroid tumors in male rats through a disruption
in the thyroid-pituitary hormonal feedback mechanisms.
-- Among scheduled and unscheduled deaths in the 78-week study,
there were significant occurrences of hepatocellular
adenomas in 52% of the males and 29% of the females, and carcinomas
in 33% of the males and 8% of the females (non-significant).
The incidences of these tumors exceeded
the corresponding historical incidence with this species in the
laboratory. Combined adenoma and carcinoma incidences at
7,000 ppm were 73% for males and 35% for
females. At 500 ppm, the incidences
of 17% adenomas and 15% carcinomas in males and 2% adenomas in
females were not statistically significant, but exceeded the means
for historical controls. The 52- and 78-week studies revealed
a dose-related decrease in the first occurrence of carcinomas
in males; the earliest carcinomas were observed
at 78, 71, 52, and 47 weeks at the 0 through 7,000 ppm doses.
There were no carcinomas in females up to 78 weeks at 0, 25, or
500 ppm, although, the earliest finding at 7000 ppm was at 60
weeks. The LOAEL for this study is 64.4 mg/kg/day for males and
77.9 mg/kg/day for females (500 ppm), based on decreased
body weight gains, increased liver
weights, and increased incidences of histopathological
liver changes. The NOEL is 3.2 mg/kg/day for males and
4.0 mg/kg/day for females (25 ppm). Although body weight was decreased
marginally in females at 25 ppm, there were no corroborating findings
of toxicity at this dose. Under conditions of this study, isoxaflutole
appears to induce hepatocellular adenomas and carcinomas in male
and female CD-1 mice. The chemical was tested at doses sufficient
to measure its carcinogenic potential.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Cholesterol
(click
on for all fluorinated pesticides)
In a combined chronic
toxicity/carcinogenicity study, isoxaflutole was continuously
administered to 75 Sprague-Dawley rats/ sex/dose at dietary levels
of 0, 0.5, 2, 20 or 500 mg/kg/day for 104 weeks. An additional
20 rats/sex/group were treated for 52 weeks, after which 10 rats/sex/group
were sacrificed and the remainder were held for a maximum of 8
weeks without treatment in order to assess reversibility of treatment-related
changes. Evidence of systemic toxicity observed at 500 mg/kg/day
in one or both sexes included: abnormal gait, limited use of limbs,
lower body weight gains and food consumption, decreased food efficiency
during the first 14 weeks of the study,
elevated cholesterol levels throughout the 104-week study,
increased absolute and relative liver weights, and thyroid hyperplasia.
Ref: Federal Register: September 23, 1998.
Isoxaflutole; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Isoxaflutole.FR.Sept23.1998.htm
Endocrine:
Pituitary
(click
on for all fluorinated pesticides)
--
Isoxaflutole demonstrates developmental toxicity and has been
classified as a Group B2 carcinogen (probable human carcinogen).
-- In a combined chronic toxicity/carcinogenicity study in rats,
evidence of systemic toxicity was observed at 500 mg/kg/day and
included: abnormal gait, limited use of limbs, lower body weight
gains and food consumption, decreased food efficiency during the
first 14 weeks of the study, elevated cholesterol levels throughout
the 104-week study, increased absolute and relative liver weights,
and thyroid hyperplasia. Increased incidence of periacinar hepatocytic
hypertrophy, portal tract (senile) bile duct changes, focal cystic
degeneration of the liver was observed in males at 20 mg/kg/day
and greater, females at 500 mg/kg/day. Eye opacity, gross necropsy
changes in eyes, corneal lesions, degeneration of sciatic nerve
and thigh muscles was observed in males at 20 mg/kg/day and higher
doses and in females at 500 mg/kg/day. The chronic LOAEL is 20
mg/kg/day based on liver, thyroid, ocular, and nervous system
toxicity in males and liver toxicity in females. The chronic NOEL
is 2.0 mg/kg/day.
-- Under the conditions of this study, isoxaflutole induced benign
and malignant tumors of the liver in both sexes at 500 mg/kg/day
hepatocellular adenomas and hepatocellular carcinomas. Combined
incidences of liver adenoma/carcinoma in males and females showed
animals bearing carcinomas in the majority. Thyroid follicular
adenomas occurred with increased frequency in 500 mg/kg/day males.
The tumor incidences exceeded the historical incidence of these
tumors for this strain in the laboratory. The study demonstrated
that isoxaflutole is carcinogenic to rats at a dose of 500 mg/kg/day.
The chemical was administered at a dose sufficient to test its
carcinogenic potential. At 500 mg/kg/day, there were alterations
in most of the parameters measured including clinical signs of
toxicity, body weight gain, food consumption, food conversion
efficiency, and clinical as well as post-mortem pathology. Thyroid
stimulating hormone (TSH) was not measured in this study. However,
in a separate special study investigating the mechanism of action
of isoxaflutole on the thyroid, tested at the same doses as this
study, TSH was indirectly measured since there was a significant
reduction in T4 level and thyroid gland weights were significantly
increased. These
results were sufficient to support the hypothesis that isoxaflutole
may have induced thyroid tumors in male
rats through a disruption in the thyroid-pituitary hormonal feedback
mechanisms.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Endocrine:
Thyroid
(click
on for all fluorinated pesticides)
-- Isoxaflutole demonstrates
developmental toxicity and has been classified as a Group
B2 carcinogen (probable human carcinogen).
-- In a combined chronic toxicity/carcinogenicity study in rats,
evidence of systemic toxicity was observed at 500 mg/kg/day and
included: abnormal gait, limited use of limbs, lower
body weight gains and food consumption, decreased food efficiency
during the first 14 weeks of the study, elevated cholesterol levels
throughout the 104-week study, increased absolute and relative
liver weights, and thyroid
hyperplasia.
Increased incidence of periacinar hepatocytic
hypertrophy, portal tract (senile) bile duct changes, focal cystic
degeneration of the liver was observed in males at 20 mg/kg/day
and greater, females at 500 mg/kg/day. Eye opacity, gross necropsy
changes in eyes, corneal lesions, degeneration of sciatic nerve
and thigh muscles was observed in males at 20 mg/kg/day and higher
doses and in females at 500 mg/kg/day. The chronic LOAEL is 20
mg/kg/day based on liver,
thyroid, ocular, and nervous system toxicity
in males and liver toxicity in females. The chronic NOEL
is 2.0 mg/kg/day.
-- Under the conditions of this study, isoxaflutole induced
benign and malignant tumors of the liver in both sexes at 500
mg/kg/day hepatocellular adenomas and hepatocellular carcinomas.
Combined incidences of liver adenoma/carcinoma in males and females
showed animals bearing carcinomas in the majority. Thyroid
follicular adenomas occurred with increased frequency in 500 mg/kg/day
males. The
tumor incidences exceeded the historical incidence of these tumors
for this strain in the laboratory. The study demonstrated
that isoxaflutole is carcinogenic to rats at a dose of 500 mg/kg/day.
The chemical was administered at a dose sufficient to test its
carcinogenic potential. At 500 mg/kg/day, there were alterations
in most of the parameters measured including clinical signs of
toxicity, body weight gain, food consumption, food conversion
efficiency, and clinical as well as post-mortem pathology. Thyroid
stimulating hormone (TSH) was not measured in this study. However,
in a separate special study investigating the mechanism of action
of isoxaflutole on the thyroid, tested
at the same doses as this study, TSH was indirectly measured since
there was a significant reduction in T4
level and thyroid gland weights were significantly increased.
These results were sufficient to support the hypothesis that isoxaflutole
may have induced thyroid tumors in male
rats through a disruption in the thyroid-pituitary hormonal feedback
mechanisms.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Eye
(click
on for all fluorinated pesticides)
-- In a combined chronic
toxicity/carcinogenicity study in rats, evidence of systemic toxicity
was observed at 500 mg/kg/day and included: abnormal gait, limited
use of limbs, lower body weight gains and
food consumption, decreased food efficiency during the first 14
weeks of the study, elevated cholesterol levels throughout the
104-week study, increased absolute and relative liver weights,
and thyroid hyperplasia. Increased incidence of periacinar hepatocytic
hypertrophy, portal tract (senile) bile duct changes, focal cystic
degeneration of the liver was observed in males at 20 mg/kg/day
and greater, females at 500 mg/kg/day. Eye
opacity, gross necropsy changes in eyes, corneal lesions, degeneration
of sciatic nerve and thigh muscles was observed in males at 20
mg/kg/day and higher doses and in females at 500 mg/kg/day.
The chronic LOAEL is 20 mg/kg/day based on liver, thyroid,
ocular, and nervous system toxicity in males
and liver toxicity in females.
The chronic NOEL is 2.0 mg/kg/day.
-- Reproductive Toxicity. In a 2-generation reproduction study
in rats, evidence of toxicity was observed in the male and female
parental rats of both generations: at 20 and 500 mg/kg/day, increased
absolute and relative liver weights associated
with liver hypertrophy was observed; at 500 mg/kg/day (HDT), decreased
body weight, body weight gain and food consumption during
premating and gestation, and increased incidence of subacute inflammation
of the cornea of the eye in F0 adults
as well as keratitis in F1 adults
were reported. There were no other systemic effects that were
attributed to treatment, nor was there any indication, at any
treatment level, of an effect on reproductive performance of the
adults. Treatment-related effects were observed in F1 and F2 offspring:
at 20 and 500 mg/kg/day, reduction in pup survival was noted;
at 500 mg/kg/day, decrease in body weights
of F1 and F2 pups throughout lactation, increased incidence of
chronic keratitis, low incidence
of inflammation of the iris, as well
as retinal and vitreous bleeding
in F2 pups and weanlings were observed. Necropsy of F1 and F2
pups culled on Day 4 revealed an increased number of pups with
no milk in the stomach and underdeveloped
renal papillae. The Systemic LOAEL is 17.4 mg/kg/day for
males and females, based upon increased
liver weights and hypertrophy and the Systemic NOEL is 1.76 mg/kg/day
for males and females. The Reproductive LOAEL is greater than
437 mg/kg/day, based on lack of reproductive effects and
the Reproductive NOEL is greater than or equal to 437 mg/kg/day.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Liver
(click
on for all fluorinated pesticides)
-- Isoxaflutole demonstrates
developmental toxicity and has been classified as a Group
B2 carcinogen (probable human carcinogen).
-- In a combined chronic toxicity/carcinogenicity study in rats,
evidence of systemic toxicity was observed at 500 mg/kg/day and
included: abnormal gait, limited use of limbs,
lower body weight gains and food consumption, decreased food efficiency
during the first 14 weeks of the study, elevated cholesterol levels
throughout the 104-week study, increased absolute and relative
liver weights, and
thyroid hyperplasia. Increased incidence of periacinar
hepatocytic hypertrophy, portal tract (senile) bile duct
changes, focal cystic degeneration of the
liver was observed in males at 20 mg/kg/day and greater,
females at 500 mg/kg/day. Eye
opacity, gross necropsy changes in eyes, corneal lesions, degeneration
of sciatic nerve and thigh muscles was observed in males at 20
mg/kg/day and higher doses and in females at 500 mg/kg/day.
The chronic LOAEL is 20 mg/kg/day based on liver,
thyroid, ocular, and nervous system toxicity in males and
liver toxicity in females. The chronic NOEL is 2.0 mg/kg/day.
-- Under the conditions of this study, isoxaflutole induced benign
and malignant tumors of the liver in both sexes at 500 mg/kg/day
hepatocellular adenomas and hepatocellular carcinomas.
Combined incidences of liver adenoma/carcinoma in males and females
showed animals bearing carcinomas in the
majority...
-- In a 78-week carcinogenicity study, isoxaflutole had no significant
effect on the survival of animals. Systemic signs of toxicity
in the treated groups included: decreased
body weight gain in both sexes at 500 ppm and 7,000 ppm and for
females at 25 ppm group; food consumption was unaffected
except food efficiency was lower for both sexes at 7000 ppm during
the first 14 weeks of the study; absolute and relative/body liver
weights were
significantly increased in both sexes at 7,000 ppm and at 500
ppm relative liver weight was increased
in males at 52 weeks and in females at 78 weeks; gross
necropsy at 78-week sacrifice revealed increased occurrences of
liver masses in both sexes at 7,000
ppm; non-neoplastic lesions of the liver
occurred at 52-week sacrifice in males at 500 ppm and in males
and females at 7,000 ppm. At termination, the 500 ppm group males
exhibited increased incidence of hepatocyte
necrosis. At 7,000 ppm, significant
increase in non-neoplastic lesions in both sexes included periacinar
hepatocytic hypertrophy, necrosis, and erythrocyte-containing
hepatocytes. In addition, males at the high dose had pigment-laden
hepatocytes and Kupffer cells, basophilic foci, and increased
ploidy; extramedullary hemopoiesis in the spleen was noted in
both sexes; increase incidences of hepatocellular
adenoma and carcinoma were observed in both sexes at 7,000
ppm in the 52-week and 78-week studies.
-- Among scheduled and unscheduled deaths in the 78-week study,
there were significant occurrences of hepatocellular
adenomas in 52% of the males and 29% of the females, and carcinomas
in 33% of the males and 8% of the females (non-significant).
The incidences of these tumors exceeded
the corresponding historical incidence with this species in the
laboratory. Combined adenoma and carcinoma incidences at
7,000 ppm were 73% for males and 35% for
females. At 500 ppm, the incidences
of 17% adenomas and 15% carcinomas in males and 2% adenomas in
females were not statistically significant, but exceeded the means
for historical controls. The 52- and 78-week studies revealed
a dose-related decrease in the first occurrence of carcinomas
in males; the earliest carcinomas were observed
at 78, 71, 52, and 47 weeks at the 0 through 7,000 ppm doses.
There were no carcinomas in females up to 78 weeks at 0, 25, or
500 ppm, although, the earliest finding at 7000 ppm was at 60
weeks. The LOAEL for this study is 64.4 mg/kg/day for males and
77.9 mg/kg/day for females (500 ppm), based on decreased
body weight gains, increased liver
weights, and increased incidences of histopathological
liver changes. The NOEL is 3.2 mg/kg/day for males and
4.0 mg/kg/day for females (25 ppm). Although body weight was decreased
marginally in females at 25 ppm, there were no corroborating findings
of toxicity at this dose. Under conditions of this study, isoxaflutole
appears to induce hepatocellular adenomas
and carcinomas in male and female CD-1 mice. The chemical
was tested at doses sufficient to measure its carcinogenic potential.
-- Reproductive Toxicity. In a 2-generation reproduction study
in rats, evidence of toxicity was observed in the male and female
parental rats of both generations: at 20 and 500 mg/kg/day, increased
absolute and relative liver weights
associated with liver hypertrophy
was observed.. The Systemic LOAEL is 17.4 mg/kg/day for males
and females, based upon increased liver
weights and hypertrophy and the Systemic NOEL is 1.76 mg/kg/day
for males and females...
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Lymph
node (click
on for all fluorinated pesticides)
An oncogenicity study
in mice administered 0, 25, 500 and 7,000 ppm with a NOEL of 25
ppm based on a slight effect on liver weight and body weight gain
at the LEL of 500 ppm. An increased incidence of hepatocellular
adenomas and carcinomas was observed at 7,000 ppm in both sexes.
Increased liver weight, non-neoplastic cellular changes in the
liver, and amyloidosis in the duodenum,
ileum, jejunum, kidneys, heart ventricle, mesenteric
lymph node, and thyroid were also observed at 7,000 ppm.
Ref: Federal Register: February 26, 1997
(Volume 62, Number 38)] [Notices] [Page 8737-8740]. Rhone-Poulenc
Ag Company; Pesticide Tolerance Petition Filing.
http://www.epa.gov/EPA-PEST/1997/February/Day-26/p4628.htm
Sciatic
nerve (click
on for all fluorinated pesticides)
-- In a combined chronic
toxicity/carcinogenicity study in rats, evidence of systemic toxicity
was observed at 500 mg/kg/day and included: abnormal gait, limited
use of limbs, lower body weight gains and
food consumption, decreased food efficiency during the first 14
weeks of the study, elevated cholesterol levels throughout the
104-week study, increased absolute and relative liver weights,
and thyroid hyperplasia. Increased incidence of periacinar hepatocytic
hypertrophy, portal tract (senile) bile duct changes, focal cystic
degeneration of the liver was observed in males at 20 mg/kg/day
and greater, females at 500 mg/kg/day. Eye opacity, gross necropsy
changes in eyes, corneal lesions,
degeneration of sciatic nerve and thigh
muscles was observed in males at 20 mg/kg/day and higher doses
and in females at 500 mg/kg/day.
The chronic LOAEL is 20 mg/kg/day based on liver, thyroid, ocular,
and nervous system toxicity in males and liver toxicity in females.
The chronic NOEL is 2.0 mg/kg/day.
Ref: US EPA. Pesticide Fact Sheet. Isoxaflutole
Reason for Issuance: Conditional Registration Date Issued: September
15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
Thyroid
(click
on for all fluorinated pesticides)
Likely
to be carcinogenic to Humans. Statistically
significant increases in liver tumors
in both sexes of CD-1 mice & Sprague-Dawley rats; statistically
significant increases in thyroid
tumors in male rats.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
| Environmental
(click
on for all fluorinated pesticides)
--
The data available at this time indicate that isoxaflutole
is very phytotoxic.
-- Isoxaflutole is persistent and
mobile, and may leach and accumulate in groundwater and
through surface water.
-- Potential to Contaminate Groundwater.
Isoxaflutole is mobile and is expected
to persist and accumulate in surface water and groundwater.
Modeling data show that parent isoxaflutole and its
primary metabolite RPA 202248 may accumulate to concentrations
that would result in harm to non-target plants. IsoxaflutoleÕs
terminal metabolite RPA 203328 is expected to persist and
accumulate, but does not demonstrate phytotoxicity. Additional
studies, including prospective groundwater studies and surface
water monitoring, will be conducted to determine whether
isoxaflutole and its primary metabolite RPA 202248 do or
do not exceed concentrations deemed potentially harmful
to the environment.
-- Aquatic - Freshwater. Isoxaflutole is moderately
toxic to the rainbow trout (96-hour LC50 > 1.7 ppm)
and to the bluegill sunfish (96-hour LC50 > 4.5 ppm). It
is also moderately toxic to Daphnia
magna (48-hour EC50 > 1.5 ppm).
-- Aquatic - Estuarine/Marine. Isoxaflutole is
highly toxic to the mysid shrimp (96-hour LC50 /EC50
= 0.018 ppm) and moderately toxic to the eastern oyster
(96-hour LC50 /EC50 = 3.3 ppm). It is moderately toxic to
the sheepshead minnow (96-hour LC50 > 6.4 ppm)
-- Plants. Isoxaflutole is highly
toxic terrestrial plants (EC25 = 1 X 10 -5 pounds
active ingredient./Acre). Due to the low vapor pressure
of this herbicide, and due to the fact that it is only to
be applied using ground equipment, risk to nontarget plant
species is not expected from the parent compound. The primary
metabolite RPA 202248, however, is mobile and is expected
to move off-site. Additional studies, including prospective
groundwater studies and surface water monitoring, will be
conducted to determine whether isoxaflutole and its primary
metabolite RPA 202248 do or do not exceed concentrations
deemed potentially harmful to the environment.
Ref: US EPA. Pesticide Fact
Sheet. Isoxaflutole Reason for Issuance: Conditional Registration
Date Issued: September 15, 1998.
http://www.epa.gov/opprd001/factsheets/isoxaflutole.pdf
|
| A
February
16, 2005,
check at the Code
of Federal Regulations for Isoxaflutole: this herbicide
is permitted in or on 28
food
commodities in the United States.
The
following list identifies these crops for which EPA has set
pesticide tolerances. |
| [Code
of Federal Regulations]
[Title 40, Volume 22]
[Revised as of July 1, 2004]
From the U.S. Government Printing Office via GPO Access
[CITE: 40CFR180.537]
[Page 495-496]
TITLE 40--PROTECTION OF ENVIRONMENT
CHAPTER I--ENVIRONMENTAL PROTECTION AGENCY (CONTINUED)
PART 180_TOLERANCES AND EXEMPTIONS FROM TOLERANCES FOR PESTICIDE
CHEMICALS
IN FOOD--Table of Contents
Subpart C_Specific Tolerances
Sec. 180.537 Isoxaflutole; tolerances for residues.
(a) General. (1) Tolerances are established for
combined residues of
the herbicide isoxaflutole [5-cyclopropyl-4-(2-methylsulfonyl-4-
trifluoromethyl benzoyl) isoxazole] and
its metabolites 1-(2-
methylsulfonyl-4-trifluoromethylphenyl)-2-cyano-3-cyclopropyl
propan-
1,3-dione (RPA 202248) and 2-methylsulphonyl-4-trifluoromethyl
benzoic
acid (RPA 203328), calculated as the parent compound, in or
on the
following raw agricultural commodities: |
| Commodity |
As
of
October 11, 2003
PPM |
As
of
February 16, 2005
PPM |
CFR |
| Corn, field,
forage |
1.0 |
1.0 |
180.537
|
| Corn, field,
grain |
0.20 |
0.20 |
180.537
|
| Corn, field,
stover |
0.50 |
0.50 |
180.537
|
| 2)
Tolerances are established for combined
residues of the
herbicide isoxaflutole [5-cyclopropyl-4-(2-methylsulfonyl-4-
trifluoromethyl benzoyl) isoxazole] and
its metabolite 1-(2-
methylsulfonyl-4-trifluoromethylphenyl)-2-cyano-3-cyclopropyl
propan-
1,3-dione (RPA 202248), calculated as the parent compound,
in or on the
following raw agricultural commodities: |
| Cattle,
fat |
0.20 |
0.20 |
180.537
|
| Cattle,
liver |
0.50 |
0.50 |
180.537
|
| Cattle,
meat |
0.20 |
0.20 |
180.537
|
| Cattle,
meat byproducts, except liver |
0.10 |
0.10 |
180.537
|
| Egg |
0.01 |
0.01 |
180.537
|
| Goat, fat |
0.20 |
0.20 |
180.537
|
| Goat, liver |
0.50 |
0.50 |
180.537
|
| Goat, meat |
0.20 |
0.20 |
180.537
|
| Goat, meat
byproducts, except liver |
0.10 |
0.10 |
180.537
|
| Hog, fat |
0.20 |
0.20 |
180.537
|
| Hog, liver |
0.50 |
0.50 |
180.537
|
| Hog, meat |
0.20 |
0.20 |
180.537
|
| Hog, meat
byproducts, except liver |
0.10 |
0.10 |
180.537
|
| Horse,
fat |
0.20 |
0.20 |
180.537
|
| Horse,
liver |
0.50 |
0.50 |
180.537
|
| Horse,
meat |
0.20 |
0.20 |
180.537
|
| Horse,
meat byproducts, except liver |
0.10 |
0.10 |
180.537
|
| Milk |
0.02 |
0.02 |
180.537
|
| Poultry,
fat |
0.20 |
0.20 |
180.537
|
| Poultry,
liver |
0.30 |
0.30 |
180.537
|
| Poultry,
meat |
0.20 |
0.20 |
180.537
|
| Sheep,
fat |
0.20 |
0.20 |
180.537
|
| Sheep,
liver |
0.50 |
0.50 |
180.537
|
| Sheep,
meat |
0.20 |
0.20 |
180.537
|
| Sheep,
meat byproducts, except liver |
0.10 |
0.10 |
180.537
|
| (b)
Section 18 emergency exemptions. [Reserved]
(c) Tolerances with regional registrations. [Reserved]
(d) Indirect or inadvertent residues. [Reserved] |
|

