|
Return to Index
Page
ACTIVITY: Acaricide,
Insecticide (pyrrole)
CAS Name: 4-bromo-2-(4-chlorophenyl)-1-(ethoxymethyl)-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile
Structure:
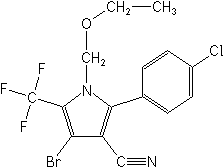
| |
| Date
Published |
Docket
Identifiction Number |
Details |
| Jan
26, 2005 |
OPP-2004-0362 |
BASF
-
Pesticide Tolerance.
FINAL
RULE.
This regulation establishes a tolerance for residues of chlorfenapyr
in or on all foods
at 0.01 parts per million (ppm) except
fruiting vegetables. •
This Final Rule contains a separate document that
is EPA's response to FAN's comments - see
http://www.fluorideaction.org/pesticides/chlorfenapyr.2005.epa.response.pdf
(published
in EPA Docket, Number: OPP-2004-0362-0002)
•
Aug 11, 2003: FAN's comments
on BASF's petition for this tolerance at
http://www.fluorideaction.org/pesticides/chlorfenapyr.comments.aug03.htm
•
Sept 17, 2003: BASF's response
to FAN's comments at
http://www.fluorideaction.org/pesticides/chlorfenapyr.basf.sept.2003.pdf
•
The nature of the toxic effects caused by chlorfenapyr are
discussed in a September 26, 2003, Final Rule - see comments
immediately below.
•
See BASF's petition (July 16, 2003) for this Tolerance at
http://www.fluorideaction.org/pesticides/chlorfenapyr.fr.july16.2003.htm
••
Conditions for this Final Rule: A
developmental neurotoxicity study to determine the
cause/relationship
of potential central nervous system/myelinopathic alterations
to neurotoxicity in the developing young. This condition was
stated in the September
26, 2003, Federal Register, and referred to in this Final
Rule.
|
|
Sept
26, 2003
|
OPP-2003-0146
|
BASF
- Pesticide Tolerance.
FINAL RULE. This regulation establishes a
tolerance for residues of chlorfenapyr at 1.0 ppm in or on
vegetables, fruiting, group 8,
grown in greenhouses. BASF
Agro Research, now BASF Corporation requested this
tolerance. In the FR of September 13, 2000, EPA announcied
the filing of an amended pesticide petition (PP 6F4716) by
BASF Agro Research, now BASF Corporation.
--
MRID No. 42770219 (1993)-- 90-Day
oral toxicity rats. NOAEL = 24.1 mg/kg/day. LOAEL =
48.4, based on spongiform
myelopathy in
the brain and spinal cord
of male rats, decreased
body weight gain and increased relative liver weight in males
and females, increased absolute liver weight in females,
and decreased hemoglobin in females.
-- MRID No. 43492830
(1994).
90-Day oral toxicity mouse
.
NOAEL = 27.6/40, M/F. LOAEL = 62.6/78, M/F, based
on reduced body weights/body
weight gains, and spongiform encephalopathy
in both sexes.
-- MRID No. 42770220 (1993).
90-Day oral toxicity dog. NOAEL
= 3.9/4.5 mg/kg/ day, M/F. LOAEL
= 6.7/6.8 mg/kg/ day, M/Fbased on emaciation,
decreased body weight gains, and decreased food efficiency.
-- MRID No. 43492831 (1993). 21/28-Day
dermal toxicity rabbit. NOAEL
= 100 mg/kg/day. LOAEL = 400 mg/kg/day, for both sexes, based
on changes in liver chemistry
and morphology.
-- MRID No. 42884202
(1993). Prenatal developmental
rat. Maternal NOAEL = 25 mg/kg/day,
based on decreased body weight
gain and relative food consumption during treatment Developmental
NOAEL >=225 mg/kg/day. Developmental LOAEL = not identified.
-- MRID No. 42770222 (1993). Prenatal
developmental rabbit.
Maternal NOAEL = 5 mg/kg/day.
Maternal LOAEL = 15 mg/ kg/day, based on decreased
body weight gain during treatment Developmental NOAEL
= 15 mg/kg/day Developmental LOAEL = 30 mg/kg/day, based on
increased post implantation loss.
-- MRID No. 43492836 (1994). 2-Generation
reproduction and fertility effects rat. Parental
systemic NOAEL = 4.4-4.5 mg/kg/day, M.
Parental systemic LOAEL = 22.2-22.5 mg/kg/day, M,
based on decreased absolute body
weight/body weight gains of P1 males
during premating. Offspring
systemic NOAEL = 4.4-5.1 mg/kg/day.
Offspring systemic LOAEL = 22.2-25.6 mg/kg/day, based
on decreased pup weights at weaning.
Reproductive NOAEL >=44- 50.7 mg/kg/day.
Reproductive LOAEL: not identified.
-- MRID No. 43492834 (1994).
Chronic toxicity dog.
NOAEL = 4.0/4.5 mg/kg/day, M/F.
LOAEL = 8.7/10.1 mg/kg/ day, M/F, based on decreased
body weight/body weight gains.
-- MRID No. 43492838
(1994). Carcinogenicity
mouse. NOAEL
= 2.8/3.7 mg/kg/day, M/F. LOAEL
= 16.6/21.9 mg/kg/day, M/F, based on decreased
body weight gains, brain vacuolation, and scabbing of the
skin (males) No evidence
of carcinogenicity.
-- MRID No. 43492837
(1994). Combined chronic/ carcinogenicity
in rat.
NOAEL = 15 mg/kg/day, males. LOAEL =
30.8 mg/kg/day, males, based
on anemia. NOAEL
= 3.6 mg/kg/day, females LOAEL = 18.6 mg/kg/day, females,
based on decreased body weight/
body weight gain. Classification: ``Suggestive
Evidence of Carcinogenicity,
but Not Sufficient to Assess Human Carcinogenic Potential''
based on significant trends in liver
tumors (adenomas and combined adenomas/ carcinomas),
malignant histiocytic sarcomas,
and testicular cell tumors in
male rats and uterine polyps
in female rats seen at the highest dose.
--
MRID No. 43492833 (1994).
Chronic neurotoxicity rat. NOAEL
= 2.6/3.4 mg/kg/day, M/F. LOAEL
= 13.6/18 mg/kg/ day, M/F, based on the presence of
myelinopathic alterations in the central
nervous system (CNS) in male rats
and decreased average body weights/body
weight gains, food efficiency, absolute food consumption (females)
and water consumption (males)
-- Developmental toxicity study - rabbit.
LOAEL = 30 mg/kg/day based on increased post-implantation
loss
-- Acute neurotoxicity study - rat.
LOAEL = 90 mg/kg/day based on lethargy
in male rats.
-- Chronic neurotoxicity study - rat.
LOAEL = 13.6/18 mg/kg/ day, M/F, based
on the presence of myelinopathic
alterations in the CNS in male rats
and decreased average body weights,
body weigh gains, food efficiency, absolute food consumption
(F), and water consumption (M) Supporting
this endpoint are similar CNS
lesions and skin lesions observed
in the mouse carcinogenicity study (NOAEL
= 2.8).
-- The most highly exposed population
subgroup is children 1-2 years old, at 47% of the cPAD.
-- EPA concluded that a FQPA SF in the form of UFDB of 10X
is required until the data from the DNT study are received
and evaluated.
-- Conditions:
A developmental neurotoxicity study to
determine the cause/relationship of potential central nervous
system/myelinopathic alterations to neurotoxicity in the developing
young. |
| July
16, 2003 |
OPP-2003-0205 |
BASF
-
Petition for pesticide tolerance.
--
Pesticide petition (PP 3F6560) proposes to establish a tolerance
for residues of chlorfenapyr on all food items in food handling
establishments where food products are held, processed, and/or
prepared at 0.01 parts per million (ppm) due to application
of chlorfenapyr to crack and crevice and spot applications.
-- Note: the following is the opinion
of BASF, not US EPA.
-- Males appear to be more sensitive to the effects of chlorfenapyr
than females.
-- Food. There are currently no established
U.S. permanent food tolerances for chlorfenapyr. There
are two tolerance petitions pending at EPA; 0.5 ppm tolerance
on imported citrus and 1.5 ppm tolerance on greenhouse grown
vegetable, fruiting, crop group 8. A dietary exposure
estimate based on theoretical maximum residue contribution
(TMRC) was conducted using the Dietary Exposure Evaluation
Model (DEEMTM) The TMRC is a ``worst case'' estimate for dietary
exposure because it assumes that 100% of crop is treated and
residues in the food are always found at the tolerance level.
Additional assumptions used were all consumption of tomatoes-whole
is from treated greenhouse grown tomatoes, greenhouse grown
tomatoes are not processed, and all citrus juice in the U.S.
is made from treated imported citrus pulp. Default processing
factors were used to determine concentrations in processed
fractions. The tolerance levels used in the dietary assessment
were 0.5 ppm for citrus pulp, 1.5 ppm for vegetable, fruiting,
group 8, and 0.01 ppm for all other crops.
-- Approved uses in the U.S. include
applications to ornamental plants inside greenhouses, to a
narrow band of soil adjacent to buildings and to crack-and-crevice
and spot treatments inside structures. A pending use expands
greenhouse applications to vegetable, fruiting, crop group
-- The parent molecule, chlorfenapyr is a pro-insecticide
that is converted to the active form, CL 303,268, via rapid
metabolism by mixed function oxidases (MFOs). The
active form uncouples oxidative phosphorylation in the insect
mitochondria by disrupting the proton gradient across the
mitochondrial membrane. The production of ATP is inhibited
resulting in the cessation of all cellular functions. Because
of this unique mechanism of action, it is highly unlikely,
that toxic effects produced by chlorfenapyr would be cumulative
with those of any other pesticide chemical.
-- In mammals, there is a lower titer of MFOs, and chlorfenapyr
is metabolized by different pathways (including dehalogenation,
oxidation and ring hydroxylation) to other polar metabolites
without any significant accumulation of the potent uncoupler,
CL 303,268.
-- Other than children less than
12 years of age, hispanics are the U.S. population subgroup
with the highest chronic exposure of 0.003403 mg/kg bwt/day,
or 11.3% of the RfD.
-- In
Mexico there is a MRL of 0.3 ppm for cottonseed.
NOTE:
see comments submitted
by FAN to US EPA on August 11, 2003; and for a response
to these comments by BASF
on September 17, 2003. |
| May
17, 2002 |
OPP-2002-0037 |
Revocation
of Expired Time Limited Tolerances. FINAL RULE .
Time-limited tolerances for fat; meat byproducts and meat of
cattle, goats, hogs, horses and sheep; cottonseed; cotton gin
byproducts; milk; and milk fat are being removed from 40 CFR
180.513 because they expired on January 31, 2001. |
| Jan
25, 2002 |
na |
BASF
-
Extension
of Experimental Use Permit ( 241-EUP-141) effective from
Nov 26, 2001 to Dec 31, 2002. This EUP allows the use of 289.27
pounds of the termiticide chlorfenapyr (4-bromo-2-(4-chlorophenyl)-1-(ethoxymethyl)-
5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile) on less than
22 acres of residential/commercial structures to evaluate
the control of termites. The program is authorized only in
the States of AL, AZ, AR, CA, CT, DE, DC, FL, GA, HI, ID,
IL, IN, IO, KY, LA, MD, MA, MI, MS, MO, NE, NJ, NY, NC, OH,
OR, PA, RI, SC, TN, TX, VA, and WA. |
| Mar
19, 2001 |
OPP-30485A/30464B |
BASF
(formerly American Cyanamid)
- Product
Approvals for Alert (Pylon); AC 303,630 Technical (Chlorfenapyr
Technical). |
| Sept
13, 2000 |
PF-971 |
BASF
- Pesticide tolerance
petition; in or on the raw agricultural commodity (RAC)
(fruiting vegetables grown under greenhouse conditions) at 1.5
ppm.
-- Analytical method. The gas chromatography
(GC) analytical method, M 2427, which is proposed as the enforcement
method for the residue of chlorfenapyr in tomato and pepper,
has a limit of quantitation (LOQ) of 0.05
ppm.
-- Magnitude of residues. Tomato
and pepper field trials have been conducted. The residue data
were collected from studies having multiple applications (5)
of chlorfenapyr with a maximum seasonal rate of up to 0.60 -
1.0 lb active ingredient/Acre to tomato and pepper. The resulting
chlorfenapyr residues in the raw agricultural commodity ranged
from <0.05 ppm (the LOQ of the method) to 1.2 ppm.
--
Males appear to be more sensitive to the
effects of chlorfenapyr than females.
-- Rat oral teratology. NOAEL for maternal toxicity 25 mg/kg
bwt/ day and NOAEL for fetal/developmental toxicity at 225 mg/kg
bwt/day.
-- Rabbit
oral teratology. NOAEL for maternal 5 mg/kg bwt/day and NOAEL
for fetal/developmental toxicity 30 mg/kg bwt/day
-- Rat 2-generation reproduction. NOAEL for parental toxicity/
growth and offspring development 60 ppm (5 mg/kg bwt/day) and
NOAEL for reproductive performance 600 ppm (44 mg/kg bwt/day).
-- Chronic toxicity tests: 1-year
neurotoxicity in rats. NOAEL 60 ppm (2.6/3.4 mg/kg bwt/ day
MF).
-- A petition for a permanent tolerance
at 0.5 ppm for imported citrus is pending at the Agency.
|
| May
10, 2000 |
OPP-34162 |
AMERICAN
CYANAMID
withdraws
Application to Register Products used on Cotton. On March
13, 2000, Susan H. Wayland, Acting Assistant Administrator
for the Office of Prevention, Pesticides and Toxic Substances,
signed a decision memorandum for the denial of chlorfenapyr
use on cotton. |
| Nov
19, 1999 |
OPP-
30485 |
AMERICAN
CYANAMID
- Registration of
Pesticide Product: Alert insecticide miticide. Active
ingredient at 21.4%. Proposed classification/Use: For use
on ornamental crops grown in commercial greenhouses to control
spider mites, including two-spotted spider mite; worm pests,
including beet armyworm, cabbage looper, and soybean looper;
thrips, including western flower thrips; and greenhouse whiteflies.
|
| Oct
20, 1999 |
OPP-
30483 |
AMERICAN
CYANAMID
- Registration application
for pesticide product: Chlorfenapyr Insecticide Cattle Ear
Tags. Active Ingredient at 30%. Proposed classification/Use:
For use to control horn flies and lice on cattle. |
| Sept
1, 1999 |
OPP-300910 |
Re-Establishment
of Tolerances for Emergency Exemptions; for residues of
the insecticide chlorfenapyr and its metabolites in or on cottonseed
and cotton gin byproducts at 0.5 and 2.0 ppm, respectively,
and in livestock commodities at levels ranging from 0.01 to
0.3 ppm, for an additional 1\1/2\-year period. These tolerances
will expire and are revoked on January 31, 2001. |
| Aug
26, 1999 |
PF-880 |
AMERICAN
CYANAMID
- Petition for Pesticide
Tolerances for residues in or on imported citrus at 0.5 ppm.
As citrus processed commodities fed to food animals may
be transferred to milk and edible tissues, tolerances are
also proposed for the following ruminant food items, milk
at 0.01 ppm; milk fat at 0.15 ppm; meat at 0.01 ppm; and meat
byproducts (including fat) at 0.10 ppm. |
| Aug
25, 1999 |
na |
AMERICAN
CYANAMID
- Issuance of
Experimental Use Permit: 241-EUP-141. This EUP allows
the use of 1,600 pounds of the termiticide chlorfenapyr on
463 sites (structures) to evaluate the control of termites
and other wood boring insects. The program is authorized only
in the States of AL, AZ, AR, CA, CTt, DE, District of Columbia,
FL, GA, HI, ID, IL, IN, IO, KY, LA, MD, MA, MI, MS, MO, NE,
NJ, NY, NC, OH, OR, PA, RI, SC, TN, TX, VA, and WA. The experimental
use permit is effective from June 30, 1999 to November 30,
2000. |
| Feb
23, 1999 |
OPP-34162 |
AMERICAN
CYANAMID
- Availability of
Risk and Benefit Assessments on new pesticide registratios
of: PIRATE and ALERT for use on cotton. Extension of Comment
Period. |
| Feb
3, 1999 |
OPP-30467 |
AMERICAN
CYANAMID
- Application to
register new pesticide: Chlorfenapyr 25 WP Termiticide/Insecticide;
containing active ingredient chlorfenapyr at 25.00%. Chlorfenapyr
25 WP is intended for use by Pest Management Professionals
as a spot or crack and crevice spray for residual pest control
of termite infestations in and around houses, apartments or
other residential structures or commercial, institutional
and warehousing establishments (such as schools, supermarkets,
restaurants, and other areas). |
| Jan
20, 1999 |
OPP-34162 |
Availability
of Risk and Benefit Assessments. |
| Dec
2, 1998 |
OPP-30464 |
AMERICAN
CYANAMID
- Applications to
register 3 Pesticide Products: AC 303,630 Technical (for technical
manufacturing); and for restricted use on cotton: Alert and
Pirate (both Insecticide/Miticide). |
| Sept
30, 1998 |
PF-828 |
AMERICAN
CYANAMID
- Pesticide Tolerance
Petition; for residues in or on the raw agricultural commodity
milk, milk fat, meat, meat fat and meat byproducts at 0.01,
0.03, 0.01, 0.03, and 0.30 ppm respectively, derived from
the use of chlorfenapyr ear tags on beef and dairy cattle.
|
|
June 15, 1998 |
OPP-30455 |
AMERICAN
CYANAMID
- Application to
register the pesticide product: Chlorfenapyr Termiticide-Insecticide.
Active ingredient Chlorfenapyr at 21.44 %. Proposed classification/Use:
General. For use on cockroaches, ants, and wood infesting
insects. |
| March
27, 1998 |
PF-799 |
AMERICAN
CYANAMID
- Pesticide
Petition for a tolerance of 0.5 ppm for residues in or on
the raw agricultural commodity citrus. As citrus processed
commodities fed to food animals may be transferred to milk
and edible tissues, tolerances are also proposed for the following
ruminant food items: milk at 0.01 ppm; milk fat at 0.15 ppm;
meat at 0.01 ppm; and meat by-products (including fat) at
0.10 ppm. |
| Feb
4, 1998 |
na |
AMERICAN
CYANAMID
- Issuance of 2 Experimental
Use Permits:
-- 241-EUP-142 - allows the use
of 42.92 pounds of the insecticide chlorfenapyr on 2,160 head
of cattle to evaluate the control of horn flies and lice.
The program is authorized only in the States of AL, AR, CA,
FL, GA, ID, IN, KS, KY, LA, MD, MS, MO, NE, NM, NC, ND, OK,
TX, WI, and WY. The EUP is effective from December 23, 1997
to July 31, 1998. Time- limited tolerances have been established
for residues of the active ingredient in or on cattle (fat,
mbyp, and meat).
-- 241-EUP-143 - allows the use
of 21.33 pounds of the insecticide chlorfenapyr on 2,160 head
of cattle to evaluate the control of horn flies and lice.
The program is authorized in the states cited above. The experimental
use permit is effective from December 23, 1997 to July 31,
1998.
--Both
permits will use the same active ingredient but different
formulations. |
| Aug
22, 1997 |
OPP-300529 |
Pesticide
Tolerances for Emergency Exemptions. - FINAL RULE. This
regulation establishes time-limited tolerances for chlorfenapyr
in or on cottonseed; cotton gin byproducts; milk; milk fat;
meat of cattle, goats, hogs, horses, and sheep; fat of cattle,
goats, hogs, horses, and sheep; and meat byproducts of cattle,
goats, hogs, horses and sheep. These tolerances will expire
and are revoked on July 31, 1999. |
| June
13, 1997 |
na |
Emergency Exemption Request Withdrawn: Arizona Department
of Agriculture withdrew their specific exemption request for
the use of chlorfenapyr on lettuce to control the beet armyworm
on December 27, 1996. |
| Mar
5, 1997 |
OPP-181033 |
Application
for Emergency Exemption. Request from the Texas Department
of Agriculture to use the pesticide chlorfenapyr, formulated
as Pirate 3SC, to treat up to 1.8 million acres of cotton to
control the beet armyworm. |
| Feb
5, 1997 |
PF-697 |
AMERICAN
CYANAMID
- Pesticide Tolerance
Petition of 0.5 ppm in or on cottonseed. |
| Nov
13, 1996 |
na |
PIRATE.
Emergency Exemption for use on cotton in TN. |
| Oct
30, 1996 |
OPP-181027 |
Request
for Emergency Exemption to treat up to 65,000 acres of lettuce
in AZ. |
| Oct
9, 1996 |
na |
PIRATE.
Emergency Exemption for use on cotton in GA. |
| Oct
2, 1996 |
OPP-181022 |
PIRATE.
Request for Emergency Exemption for use on cotton in CA. |
| July
24, 1996. |
OPP-181018 |
PIRATE.
Request for Emergency Exemption to treat up to 1 million acres
of cotton in TN. |
| June
19, 1996 |
na |
PIRATE.
Emergency Exemption for use in LA. |
| June
12, 1996 |
PF-646 |
AMERICAN
CYANAMID
- Pesticide Tolerance
Petition to add an import tolerance of 0.5 ppm for residues
of AC 303,630. |
| May
10, 1996 |
na |
PIRATE.
Emegency Exemption for use on cotton in Texas. |
| April
10, 1996 |
OPP-181006 |
PIRATE.
Request for Emergency Exemption to treat up to 3,300,000 acres
of cotton and tobacco in Alabama and Mississippi. |
| Mar
20, 1996 |
OPP-181001 |
PIRATE.
Request from Louisiana for Emergency Exemption to treat up to
1 million acres of cotton. |
| Feb
28, 1996 |
OPP-180991 |
Request
for Emergency Exemption to treat up to 1.8 million acres of
cotton in TX. |
| Feb
21, 1996 |
na |
PIRATE.
Emergency Exemptions for use on cotton in AL, AR, FL, GA, LA,
MS, TX. |
| Sept
13, 1995 |
OPP-180980 |
PIRATE.
Emergency Exemption for use on cotton in AL, AR, LA, MS. |
| July
7, 1995 |
OPP-180974 |
PIRATE.
Requests for Emergency Exemption for use on up to 2,125,000
acres in the southeastern region of the cotton belt (AL, AR,
LA, MS, TN). |
| Mar
29, 1995 |
na |
AMERICAN
CYANAMID
- Issuance of Experimental
Use Permit (241-EUP-128) for use on 150 acres of greenhouse
and shadehouse ornamentals to evaluate the control of various
insect pests. The EUP is effective from January 18, 1995 to
January 18, 1997. |
|