Abstract
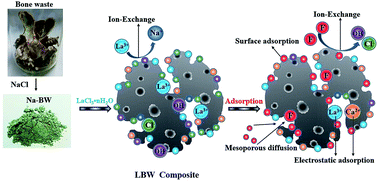
The current worldwide issue of fluoride contamination in groundwater has resulted in an increased demand for efficient adsorbents. Meanwhile discard and landfill of bone waste has led to environmental pollution. In order to achieve bone waste recycling and enhance the efficiency of fluoride removal, a lanthanum-modified bone waste (LBW) composite was synthesized and tested to remove fluoride from contaminated groundwater. The adsorbent characterization was conducted by SEM, BET, XRD, FTIR and XPS. The fluoride adsorption performance was evaluated by batch experiments. SEM and BET revealed that the introduction of lanthanum could modify the porous structure of the adsorbent and enhance its specific surface area. The LBW composite had a high pHzpc of 11.4 and the fluoride adsorption was barely affected by the solution pH over a wide pH range of 2.5–10.0. The influence of common co-existing oxygen anions in the range of 0–100 mg L?1 was not significant. The fluoride adsorption was a typical chemisorption process and followed PSO and F-L PSO equations. The FVER model provided a more accurate prediction of a larger surface coverage degree with respect to equilibrium at the initial stage during adsorption of fluoride. Isotherm studies revealed that the reaction obeyed the Langmuir model, indicating that this process was monolayer adsorption. Possible defluoridation and regeneration were proposed. The fluoride adsorption was mainly controlled by the processes of electrostatic attraction on the LBW surface with a positive charge and ion exchange between fluoride and hydroxide ions. This research provides an alternative method for fluoride removal from contaminated groundwater in practical applications.
*FULL ARTICLE online at http://pubs.rsc.org/en/content/articlehtml/2017/ra/c7ra10713g
