Abstract
Highlights
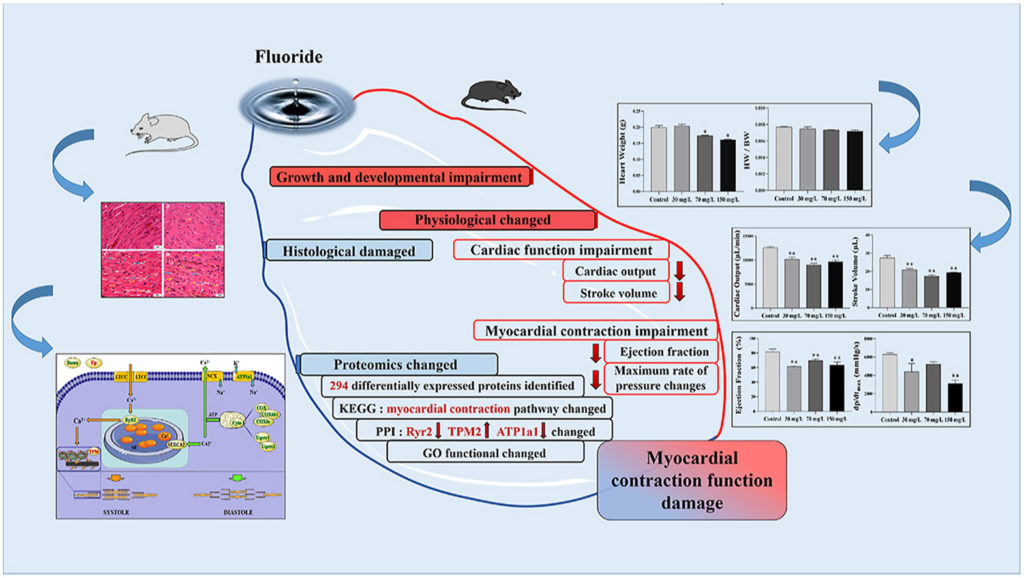
- Pressure-volume loop analysis highlighted the impaired effects of fluoride exposure on cardiac function and myocardial contraction indicators.
- Histological analysis of rat heart tissue under fluoride exposure revealed extensive damage.
- Proteomics analysis highlighted effects on the myocardial contraction pathway.
- Protein network analysis highlighted the key roles of RyRs, Tpm and AT1A1.
Fluorosis is a worldwide public health problem, and its adverse effects on the heart have been confirmed by many studies. Abnormal myocardial contractions are often associated with impairment of cardiac function as a cause or consequence. We designed two-part experiments to search for biomarkers and clarify the underlying molecular mechanism of fluoride on myocardial contraction. First, we used Pressure-volume Loop analysis to evaluate changes in myocardial function indexes with multiple fluoride exposure levels in mice (0, 30, 70, and 150 mg/L) exposed for 4 weeks. The results showed that fluoride exposure affects the heart pump function and reduces cardiac contractility. Then, we established a rat model of fluoride exposure (0, 30, 60, and 90 mg/L) for 6 months to carry out proteomic analysis of fluoride-induced myocardial contractile injury. Hematoxylin-eosin (H&E) staining was used to determine the severity of myocardial injury, and myocardial tissue samples were submitted for isobaric tags for relative and absolute quantitation (ITRAQ) analysis. A total of 1607 proteins were successfully identified with 294 differentially expressed proteins (DEPs) in fluoride treated groups. According to Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis, 12 DEPs were confirmed to be involved in pathways related to myocardial contraction. Furthermore, we constructed a protein-protein interaction (PPI) network for these 12 core DEPs to illustrate the role and location of each DEP in the myocardial contraction pathway. The results of this study are helpful for identify a potential mechanism and biomarkers of fluoride-induced myocardial contraction function damage, moreover, which can provide a new insight into the heart toxicity of fluoride in animals at the proteomics level.
Graphical abstract
*Original abstract online at: https://www.sciencedirect.com/science/article/abs/pii/S0147651320304449?via%3Dihub
