|
Return
to Index Page
Activity:
Herbicide
(pyrazolylphenyl)
Structure
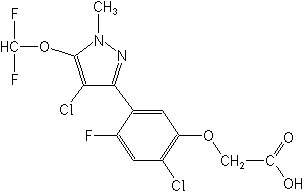
for "Pyraflufen"
Adverse Effects:
Body Weight Decrease
Blood
Cancer: Likely to be
Carcinogenic to Humans: LIVER
Eye
Kidney
Liver
Environmental
| •
On April 30, 2003, US EPA approved the first-time use of Pyraflufen
and Pyraflufen-ethyl in or on food commodities - see list
at bottom of the page.
•
Pyraflufen-ethyl is also registered in the US for use in/on:
Cotton, airports, nurseries, ornamental turf, golf courses,
roadsides, and railroads. |
Body
Weight Decrease (click
on for all fluorinated pesticides)
Developmental toxicity- rabbit. LOAEL = 60 mg/kg/day based on
decreases in body weight and food
consumption, GI observations, and abortions.
Ref: Federal Register: April 30, 2003. Pyraflufen-ethyl;
Pesticide Tolerance. Final Rule.
http://www.fluorideaction.org/pesticides/pyraflufen-ethy.fr.apr30.03.htm
Reproductive toxicity
Target / critical effect - Reproduction: Reduced
body weight gain of pups during lactation at parental toxic
doses. Lowest relevant reproductive NOAEL / NOEL: NOAELsyst.tox
= 1000 ppm (70.8 mg/kg bw/d) NOAELreprotox = 1000ppm ( 70.8 mg/kg
bw/d) Target / critical effect - Developmental toxicity: Implantation
loss and retardations in rabbits
at maternally toxic doses (mortality). Lowest relevant developmental
NOAEL / NOEL: 20 mg/kg bw/d
Ref: July 2, 2002 - Review report for the active substance pyraflufen-ethyl.
Finalised in the Standing Committee on Plant Health at its meeting
on 29 June 2001 in view of the inclusion of Pyraflufen-ethyl in
Annex I of Directive 91/414/EEC. SANCO/3039/99-FINAL. European
Commission Health & Consumer Protection Directorate-General.
http://www.fluoridealert.org/pesticides/Pyraflufen-eth.EU.July.2002.pdf
Blood
(click on for all fluorinated pesticides)
-- Short term toxicity
Target / critical effect:
Liver,
kidney,
red blood cells. Lowest relevant
oral NOAEL / NOEL: 200 ppm (20 mg/kg bw/d) 90 day mouse (satellite
group in 78 wk study)
-- Long term toxicity and carcinogenicity Target / critical effect:
Red blood cells and liver in mice,
urinary and biliary tract in rats.
Ref: July 2, 2002 - Review report for the
active substance pyraflufen-ethyl. Finalised in the Standing Committee
on Plant Health at its meeting on 29 June 2001 in view of the
inclusion of Pyraflufen-ethyl in Annex I of Directive 91/414/EEC.
SANCO/3039/99-FINAL. European Commission Health & Consumer
Protection Directorate-General.
http://www.fluoridealert.org/pesticides/Pyraflufen-eth.EU.July.2002.pdf
Cancer:
Likely to be Carcinogenic to Humans (click
on for all fluorinated pesticides)
Likely
to be Carcinogenic to Humans. Hepatocellular adenomas
and combined adenomas, carcinomas and/or hepatoblastomas
in male and female (SPF) ICR (Crj:CD-1) mice.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
--
Classification:
``Likely to be Carcinogenic to Humans'' by the oral route Q1*
= 3.32 x 10-2 (mg/kg/day)-1
-- Carcinogenicity
in mice. NOAEL = 200 ppm (20.99 mg/kg/day (M); 19.58 mg/kg/day
(F). LOAEL = 1,000 ppm (109.7 mg/kg/day (M); 98.3 mg/kg/day (F)
based on liver toxicity, hepatocellular
tumors at 5,000 ppm; possibly hemangioma/
hemangioasarcomas.
Ref:
Federal Register: April 30, 2003. Pyraflufen-ethyl; Pesticide
Tolerance. Final Rule.
http://www.fluorideaction.org/pesticides/pyraflufen-ethy.fr.apr30.03.htm
The
exposure from pyraflufen-ethyl residues in food results in a cancer
risk in the range of 1 in 1 million and is not a concern.
Ref: Federal Register: May 12, 2004. Pyraflufen-ethyl; Pesticide
Tolerance. Final Rule.
http://www.fluorideaction.org/pesticides/pyraflufen-ethyl.fr.may.04.htm
Eye
(click
on for all fluorinated pesticides)
Human Health Effects. The U.S. Environmental Protection Agency
(EPA) classifies Edict [which contains pyraflufe-ethyl] as category
I (High Toxicity) with a signal word of DANGER
because of irreversible damage to the eyes and harm if
swallowed or absorbed through the skin.
Ref: Pyraflufen. Roadside Vegetation Management
Herbicide Fact Sheet. Washington State Department of Transportation.
http://www.fluorideaction.org/pesticides/pyraflufen.fact.sheet.washington.pdf
Kidney
(click on for all fluorinated pesticides)
-- Short term toxicity
Target / critical effect:
Liver,
kidney, red blood
cells. Lowest relevant oral NOAEL / NOEL: 200 ppm (20 mg/kg bw/d)
90 day mouse (satellite group in 78 wk study)
Ref: July 2, 2002 - Review report for the
active substance pyraflufen-ethyl. Finalised in the Standing Committee
on Plant Health at its meeting on 29 June 2001 in view of the
inclusion of Pyraflufen-ethyl in Annex I of Directive 91/414/EEC.
SANCO/3039/99-FINAL. European Commission Health & Consumer
Protection Directorate-General.
http://www.fluoridealert.org/pesticides/Pyraflufen-eth.EU.July.2002.pdf
Liver
(click
on for all fluorinated pesticides)
Likely
to be Carcinogenic to Humans. Hepatocellular adenomas
and combined adenomas, carcinomas and/or hepatoblastomas
in male and female (SPF) ICR (Crj:CD-1) mice.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
-- Short term toxicity
Target / critical effect: Liver,
kidney, red blood cells. Lowest relevant oral NOAEL / NOEL: 200
ppm (20 mg/kg bw/d) 90 day mouse (satellite group in 78 wk study)
-- Long term toxicity and carcinogenicity Target / critical effect:
Red blood cells and liver in mice,
urinary and biliary tract in rats.Lowest relevant NOAEL: 200 ppm
(20 mg/kg bw/d ) 2 year mice study 400 ppm ( 20 mg/kg bw/d) 2
year rat study. Carcinogenicity: Increased incidence of hepatocellular
adenomas in mice at hepatotoxic doses, not carcinogenic
in rats. Classification and labelling not appropriate.
-- Other toxicological studies. Accumulation of porphyrins in
all organs except skin and Harderian glands.
Inhibitor of some liver P450 dependent activities; inhibitor
of catalase. Induction of liver single cell
necrosis followed by mitosis.
Ref: July 2, 2002 - Review report for the
active substance pyraflufen-ethyl. European Commission Health
& Consumer Protection Directorate-General.
http://www.fluoridealert.org/pesticides/Pyraflufen-eth.EU.July.2002.pdf
|
Environmental
(click
on for all fluorinated pesticides)
Particular
conditions to be taken into account on short term basis
by Member States in relation to the granting of authorisations
of plant protection products containing pyraflufen-ethyl
On the basis of the proposed and supported uses [herbicide
use in cereals], the following particular issues have been
identified as requiring particular and short term (within
12 months at the latest) attention from the Member States,
in the framework of any authorisations to be granted, varied
or withdrawn, as appropriate:
- Member States must pay particular attention to the protection
of algae and aquatic plants and should apply, where appropriate,
risk mitigation measures.
- The acid metabolite (designated
as E1) has a potential for leaching which might require
particular attention in vulnerable areas to ensure protection
of groundwater.
Ref:
July 2, 2002 - Review report for the active substance pyraflufen-ethyl.
Finalised in the Standing Committee on Plant Health at its
meeting on 29 June 2001 in view of the inclusion of Pyraflufen-ethyl
in Annex I of Directive 91/414/EEC. SANCO/3039/99-FINAL.
European Commission Health & Consumer Protection Directorate-General.
http://www.fluoridealert.org/pesticides/Pyraflufen-eth.EU.July.2002.pdf
|
| A
February
16, 2005,
check at the
Code of Federal Regulations for Pyraflufen-ethyl (and its
acid metabolite): this herbicide is permitted in
or on 13 food
commodities in the United States.
The
following list identifies these crops for which EPA has set
pesticide tolerances. |
| [Code
of Federal Regulations]
[Title 40, Volume 22]
[Revised as of July 1, 2004]
From the U.S. Government Printing Office via GPO Access
[CITE: 40CFR180.585]
[Page 514]
TITLE 40--PROTECTION OF ENVIRONMENT
CHAPTER I--ENVIRONMENTAL PROTECTION AGENCY (CONTINUED)
PART 180_TOLERANCES AND EXEMPTIONS FROM TOLERANCES FOR PESTICIDE
CHEMICALS
IN FOOD--Table of Contents
Subpart C_Specific Tolerances
Sec. 180.585 Pyraflufen-ethyl; tolerances for residues.
(a) General. Tolerances are established for
residues of the
herbicide pyraflufen-ethyl (ethyl 2-chloro-5-(4-chloro-5-
difluoromethoxy-1-methyl-1H-pyrazol-3-yl)-4-fluorophenoxyacetate)
and
its acid metabolite, E-1 (2-chloro-5-(4-chloro-5-difluoromethoxy-1-
methyl-1H-pyrazol-3-yl)-4- fluorophenoxyacetic acid), in or
on the
following raw agricultural commodities: |
| Commodity |
Parts
per million |
| Corn, field,
forage |
0.01 |
| Corn, field,
grain |
0.01 |
| Corn, field,
stover |
0.01 |
| Cotton,
gin byproduct |
1.5 |
| Cotton,
undelinted seed |
0.04 |
| Potato |
0.02 |
| Soybean,
forage |
0.01 |
| Soybean,
hay |
0.01 |
| Soybean,
seed |
0.01 |
| Wheat,
forage |
0.1 |
| Wheat,
grain |
0.01 |
| Wheat,
hay |
0.1 |
| Wheat,
straw |
0.01 |
| (b)
Section 18 emergency exemptions. [Reserved]
(c) Tolerances with regional registrations. [Reserved]
(d) Indirect or inadvertent residues. [Reserved] |
|

