|
Return to
Index Page
Adverse Effects
ACTIVITY:
Fungicide
(azole)
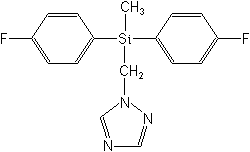
CAS Name:
1-[[bis(4-fluorophenyl)methylsilyl]methyl]-1H-1,2,4-triazole
Structure:
http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=pubmed&dopt=Abstract&list_uids=15913947&query_hl=19
Toxicol
In Vitro. 2005. Sep;19(6):737-48.
Study
on the common teratogenic pathway elicited by the fungicides
triazole-derivatives.
Menegola E, Broccia ML, Di Renzo F, Massa V, Giavini
E
Abstract:
Triazole-derivatives alter the pharyngeal apparatus
morphogenesis of rodent embryos cultured in vitro. The
hindbrain segmentation and the rhombencephalic neural
crest cell (NCCs) migration are altered by Fluconazole
exposure in vitro. The aim of the present work is to
identify if a common pathogenic pathway is detectable
also for other molecules of this class of compounds.
9.5 days post coitum (d.p.c.) old rat embryos were exposed
in vitro to the teratogenic concentrations of Flusilazole,
Triadimefon and Triadimenol and cultured for 24, 48
or 60 h. The expression and localisation of Hox-b1 and
Krox-20 proteins (used as markers for hindbrain segmentation)
were evaluated after 24 h of culture. The localisation
and distribution of NCC was evaluated after 24, 30 and
48 h of culture. The morphology of the embryos was analysed
after 48 h, while the branchial nerve structures were
evaluated after 60 h of culture. Hindbrain segmentation
and NCC migration alteration as well as pharyngeal arch
and cranial nerve abnormalities were detected after
exposure of the tested molecules. A
common severe teratogenic intrinsic property for the
tested molecules of this chemical class has been found,
acting through alteration of the normal hindbrain developmental
pattern.
|
http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=pubmed&dopt=Abstract&list_uids=15554355
Endocr
Res. 2004 Aug;30(3):387-94.
Comparative assessment of the
inhibition of recombinant human CYP19 (aromatase) by
azoles used in agriculture and as drugs for humans.
Trosken ER, Scholz K,
Lutz RW, Volkel W, Zarn JA, Lutz WK.
Department of Toxicology, University
of Wurzburg, Wurzburg, Germany.
Azoles
(imidazoles and triazoles) are used as antifungal agents
in agriculture and in medicine, and also for antiestrogen
therapy, e.g., for breast cancer treatment.
Antifungal activity is based on inhibition of fungal
CYP51 (lanosterol 14alpha-demethylase), and estrogen
biosynthesis reduction is due to azole inhibition of
CYP19 (aromatase). Inhibition of aromatase by antifungal
agents is usually an unwanted side effect and may cause
endocrine disruption. A fluorimetric assay based on
human recombinant CYP19 enzyme with dibenzylfluorescein
as a substrate was used to compare the inhibitory potency
of 22 azole compounds. Dose responses were established
and duplicate datasets were analyzed with a nonlinear
mixed-effects model with cumulative normal distribution
for the logarithm of concentration. IC50 values (50%
inhibitory concentration) of 13 fungicides used in agriculture
ranged more than 700-fold, starting from 0.047 microM.
The potency of seven human drugs spanned more than 7000-fold,
starting from 0.019 microM. Most
potent fungicides included prochloraz, flusilazole,
and imazalil, and most potent medicinal antifungals
were bifonazole, miconazole, and clotrimazole. These
in vitro data indicate that the top-ranking azoles used
as antifungal agents or drugs are as potent inhibitors
of aromatase as are antiestrogen therapeutics used to
treat breast cancer. These putative
effects of azole agents and drugs on steroid biosynthesis
and sex hormone balance should be considered when used
in human subjects and also in wildlife exposed to azole
fungicides used in agriculture.
PMID:
15554355 [PubMed - in process]
|
From
Dart Special at Toxnet
Teratology
1990 Aug;42(2):27A-28A
Triazoles
teratogenicity in rats.
Vergieva T
Inst. of Hygiene, Sofia, Bulg.
This report describes the first part of a study which
was undertaken to examine the teratogenic potential
of triazole compounds used as fungicides in agriculture.
Pregnant Wistar rats were given single oral dosages
of flusilazole or bitertanol
on days 9, 10, 11 or 13th of gestation (positive vaginal
smear=day 1) at levels of 1/5, 1/10 and 1/50 LD50. The
dosages were calculated from the reported LD50 values
of 1272 mg/kg for flusilazole and 5000 mg/kg for bitertanol.
The results of the study demonstrated that both
compounds induce congenital anomalies when given on
days 9, 10 or 11th at levels corresponding to 1/5 and
1/10 LD50. The types of the registered malformations
after flusilazole treatment were exophthalmus,
hypognathia, macroglossia and cleft palate and
after bitertanol treatment micro- and acaudia and in
rare cases exophthalmus, hypognathia and cleft palate.
A clear dose effect relationship was established for
both compounds.
• Definitions:
Exophthalmia (n.) The
protrusion of the eyeball so that the eyelids will
not cover it, in consequence of disease.
Hypognathus - Unequal
conjoined twins in which the rudimentary parasite
is attached to the mandible of the autosite.
Macroglossia - Excessively
large tongue.
|
Reports
available from
The National Technical Information Service
(NTIS)
Order from NTIS by: phone at 1-800-553-NTIS (U.S. customers);
(703)605-6000 (other countries); fax at (703)605-6900; and
email at orders@ntis.gov. NTIS is located at 5285 Port Royal
Road, Springfield, VA, 22161, USA. |
| Order
No. |
Title |
Keywords
/ Abstract |
NTIS/OTS0559123
EPA/OTS;
Doc #89980000145S |
1998
- SUPPORT: A DERMAL DOSE RANGE-FINDING PRENATAL DEVELOPMENTAL
TOXICITY STUDY OF FLUSILAZOLE IN RATS, FINAL REPORT, WITH
COVER LETTER DATED 3-6-1998
(SANITIZED)
WIL
RESEARCH LABORATORIES INC |
CONFIDENTIAL
FLUSILAZOLE
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
MAMMALS
RATS
DERMAL
CAS Registry No: 85509-19-9 |
NTIS/OTS0559123
EPA/OTS;
Doc #88970000188S |
1997
- INITIAL SUBMISSION: LETTER FROM [] TO USEPA REPORTING
PRELIMINARY RESULTS OF RAT PILOT DERMAL DEVELOPMENTAL
TOXICITY STUDY WITH FLUSILAZOLE, DATED 5-23-1997
(SANITIZED) |
CONFIDENTIAL
FLUSILAZOLE
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
MAMMALS
RATS
DERMAL
CAS Registry No: 85509-19-9 |
NTIS/OTS0538409-4
EPA/OTS;
Doc #89970000022S |
1996
- SUPPORT: LETTER FROM [ ] TO USEPA
REGARDING ACUTE ORAL TOXICITY OF A SUBSTITUTED HETEROCYCLE
AND FLUSILAZOLE, DATED 11-11-96 (SANITIZED)
|
CONFIDENTIAL
SUBSTITUTED HETEROCYCLE AND FLUSILAZOLE
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0535921
EPA/OTS;
Doc #88-920001537 |
1992
- INITIAL SUBMISSION: DEVELOPMENTAL TOXICITY STUDY
IN RATS GIVEN INF-5829 BY GAVAGE ON DAYS 7-16 OF GESTATION
(FINAL REPORT) WITH ATTACHMENTS AND COVER LETTER DATED
03-27-92
HASKELL
LAB
Benzyl
sulfonamide,N-{[(4-methoxy- 6-methyl-1,3,5-triazin-2-yl)amino]carbonyl}-2-
(propylsulfonyloxy) (CAS# 85509-19-9)
was evaluated for developmental toxicity in groups of
25 rats (Charles River CD strain) given the test substance
by oral gavage on days 7 through 16 of gestation at doses
of 0, 30, 200, and 500 mg/Kg body weight. No spontaneous
deaths occurred in any of the treatment groups. At 500
mg/Kg/day the dams exhibited decreases in food consumption
and body weight gain during the treatment period. No adverse
effects were reported in the 30 or 200 mg/Kg groups when
compared to the control group. At
the 500 and 200 mg/Kg dose level fetal toxicity was expressed
as decreased mean fetal body weight, an increased percentage
of fetuses with variations per litter, and a significant
increase in number of fetuses with unossified or partially
ossified sternebrae. The 30 mg/Kg group exhibited
fetal variations similar to controls. There were no significant
differences in sex ratios of litters between control and
treatment groups. The NOEL reported was 200 mg/KG for
the dams and 30 mg/Kg for the fetus.
Keywords:
E I DUPONT DE NEMOURS & CO
INF-5829
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0535901
EPA/OTS;
Doc #88-920001517 |
1992
- INITIAL SUBMISSION: NINETY-DAY FEEDING AND ONE-GENERATION
REPRODUCTION STUDY IN RATS WITH SILANE, [BIS(4-FLUOROPHENYL)]
(METHYL) (1H-
1,2,4-TRIAZOL-1-YLMETHYL) (INH-6573) W-ATTACH
HASKELL
LAB
(Methyl)(1H,1,2,4-Triazol-1-ymethyl-INH-6573;
11/29/83, EPA 88- 920001517, Fiche No. OTS0535901] The
subchronic toxicity of Silane (CAS# 85509-19-9)
was evaluated when fed to groups of sixteen male and sixteen
female rats (Charles River CD Strain) in a nutritionally
adequate diet at doses of 0, 25, 125, 375, and 750 ppm.
At the end of the ninety-day feeding phase, ten rats in
each group were sacrificed and processed for histopathological
examination. The remaining rats in the 0, 25, 125, and
375 ppm dose groups were used in a one-generation, one-litter
reproduction substudy to assess the effects of the test
substance on reproductive and lactation performance. Throughout
the reproduction substudy, all rats received their respective
treatment group's diet. After the last litter was weaned,
male and female parent rats were fed control diets for
four months. After this recovery phase, bladders were
removed from these rats to assess the reversibility of
lesions that had been observed in test rats sacrificed
at the end of the ninety-day feeding phase of the study.
Body weights of dams fed 125, 375,
and 750 ppm were decreased compared to controls.
Absolute and relative liver weights
of male and female rats in the high-dose group were elevated.
Relative liver weights of female rats in the 125 and 375
ppm groups were greater than controls but within
range of biological variation, therefore the 750 ppm dose
level was reported as the effect level for liver weight
change. In the reproduction substudy, the
dams at the 375 ppm dose level showed decreases in proportion
of litters with at least one live pup, percent of pups
born alive per litter, and percent of litters surviving
until weaning as compared to controls. Male
to female sex ratio of litters was not reported.
Mean litter weights were similar to controls. All
pups were sacrificed after weaning with no pathological
examination. No conclusions
regarding fertility were given as all groups, especially
that of the control and 125 ppm groups were abnormally
low. The NOEL was reported to be 125 ppm.
Keywords:
E I DUPONT DE NEMOURS & CO
INH-6573
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
RATS
ORAL
DIET
REPRODUCTION/FERTILITY EFFECTS
CAS Registry No: 85509-19-9 |
NTIS/OTS0535920
EPA/OTS;
Doc #88-920001536 |
1992
- INITIAL SUBMISSION: EMBRYO-FETAL
TOXICITY AND TERATOGENICITY STUDY OF INH-6573-39
BY GAVAGE IN THE RAT (FINAL REPORT) WITH ATTACHMENTS AND
COVER LETTER DATED 03-27-92
HASKELL
LAB
Silane,[Bis(4-Flouorophenyl)] (Methyl)(1H,1,2,4-Triazol-1-ymethyl-INH-6573
(CAS# 85509-19-9) was evaluated
to determine its embryo-fetal toxicity and teratogenic
potential when administered by oral gavage to twenty-five
rats (Charles River CD strain) per treatment group on
days 7 through 16 of gestation at doses of 0, 10, 50,
and 250 mg/kg body weight. At 250 mg/kg maternal deaths
occurred and overt toxicity expressed
as decreased body weight gain and feed consumption and
by a significant increase of chromodacryorrhea, chromorhinorrhea,
wet and stained underbodies, and alopecia. Slight
maternal toxicity at 50 mg/kg was expressed as a significant
decrease in feed consumption. The
mean relative liver weight was significantly increased
in the 250 and 50 mg/kg treatment groups. No maternal
toxicity was noted at 10 mg/kg and below. Embryo-fetal
toxicity was noted at all treatment levels. At
250 mg/kg a decrease in fetal body weight and an increase
in fetal deaths were reported. The
incidence of skeletal variations were increased significantly
above controls at all treatment levels. An
increase in fetal head malformations were reported in
all groups, including controls.
Male to female sex ratios of litters were not
reported. Due to the incidence of
fetal head malformations across all groups, no NOEL was
reported in this study.
Keywords:
E I DUPONT DE NEMOURS & CO
INH-6573-39
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0543819
EPA/OTS;
Doc #88-920006814 |
1992
- INITIAL SUBMISSION: TEN DOSE ORAL SUBACUTE TEST WITH
INH-6573-25 IN RATS WITH ATTACHMENTS, COVER SHEETS AND
LETTER DATED 08-20-92
HASKELL
LAB |
E
I DUPONT DE NEMOURS & CO
INH-6573-25
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0555076
EPA/OTS;
Doc #88-920008258 |
1992
- INITIAL SUBMISSION: SUBCHRONIC ORAL TOXICITY STUDY OF
SILANE, [BIS(4-
FLUOROPHENYL)](METHYL)(1H-
1,2,4-TRIAZOL-1-
YLMETHYL* IN RATS WITH COVER LETTER DATED
08/20/92
HASKELL
LABORATORY |
E
I DUPONT DE NEMOURS & CO
SILANE, [BIS(4-
FLUOROPHENYL)](METHYL)(1H-
1,2,4-TRIAZOL-1-YL*
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0570779
EPA/OTS;
Doc #88-920008497 |
1992
- INITIAL SUBMISSION: LETTER FROM DUPONT CHEM TO USEPA
REGARDING EMBRYO-FETAL AND TEROTOGENICITY STUDY OF SILANE*
IN RATS WITH COVER LETTER AND ATTACHMENTS DATED 08-20-92
|
DUPONT
CHEM
SILANE, [BIS(4-
FLUROPHENYL)](METHYL)(1H-1,2,4-
TRIAZOL*
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
COMBINED TERATOGENICITY/REPRODUCTIVE EFFECTS
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0570852
EPA/OTS;
Doc #88-920008564 |
1992
- INITIAL SUBMISSION: ACUTE ORAL
TOXICITY OF SILANE, [BIS(4-
FLUOROPHENYL](METHYL)(1H-
1,2,4-TRIAZOLE-1-
YLMETHYL)- IN RATS WITH COVER LETTER DATED 08-20-92
HASKELL
LABORATORY |
DUPONT
CHEM
SILANE, [BIS(4-FLUOROPHENYL)](METHYL)(1H,1,2,4-
TRIAZOLE-1-Y*
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry No: 85509-19-9 |
NTIS/OTS0570685
EPA/OTS;
Doc #88-920008403 |
1992
- INITIAL SUBMISSION: LETTER FROM
DUPONT CHEM TO USEPA REGARDING A 4-WEEK AND 90-DAY FEEDING
STUDY OF SILANE, [BIS(4-FLUROPHENYL)](METHYL)* IN MICE
WITH COVER LETTER DATED 08-20-92 |
DUPONT
CHEM
SILANE, [BIS(4-FLUROPHENYL)](METHYL) 1H-
1,2,4*
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
MICE
ORAL
DIET
CAS Registry No: 85509-19-9 |
NTIS/OTS0570690
EPA/OTS;
Doc #88-920008408 |
1992
- INITIAL SUBMISSION: LETTER FROM DUPONT CHEM TO USEPA
REGARDING 1H-1,2,4-
TRIAZOLE, 1-
(BIS-(FLURPHENYL)METHYL IN RATS WITH COVER LETTER AND
ATTACHMENTS DATED 08-20-92 |
DUPONT
CHEM
1H-1,2,4-TRIAZOLE, 1-(BIS-
(FLUORPHENYL)METHYL
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
INHALATION
CAS Registry No: 85509-19-9 |
NTIS/OTS0543806
EPA/OTS;
Doc #88-920006801 |
1992
- INITIAL SUBMISSION: INHALATION MEDIAN LETHAL CONCENTRATION
(LC50) OF INH-6573-43 BY EPA PROCOTOL WITH ATTACHMENTS,
COVER SHEET & LETTER
HASKELL
LAB |
E
I DUPONT DE NEMOURS & CO
INH-6573-43
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
INHALATION
CAS Registry No: 85509-19-9 |
NTIS/OTS0570678
EPA/OTS;
Doc #88-920008396 |
1992
- INITIAL SUBMISSION: ACUTE ORAL TOXICITY OF N7872-25
IN MALE AND FEMALE RATS WITH COVER LETTER DATED 08-20-92
HASKELL
LABORATORY |
DUPONT
CHEM
N7872-25
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
CAS Registry Nos:
10605-21-7
85509-19-9 |
NTIS/OTS0570781
EPA/OTS;
Doc #88-920008499 |
1992
- INITIAL SUBMISSION: LETTER FROM DUPONT CHEM TO USEPA
REGARDING 90-DAY FEEDING STUDY AND ONE-GENERATION REPRODUCTION
STUDY IN RATS WITH COVER LETTER AND ATTACHMENTS DATED
08-20-92 |
DUPONT
CHEM
SILANE, [BIS(4-
FLUROPHENYL)](METHYL)(1H,2,4-
TRIAZOL*
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
RATS
ORAL
DIET
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
CAS Registry No: 85509-19-9
|
Other
studies performed for US EPA:
MRID
44302410. O'Neill, A. (1994) Inhalation Median Lethal
Concentration (LC50) Study with DPX-JE874-158 in Rats.
DuPont Haskell Laboratory Report HLR 791-93.
MRID
40042109. Turner, R. (1985) Inhalation Median Lethal Concentration
(LC50) of INH- 6573. DuPont Haskell Laboratory Report
HLR 1-85.
MRID
40042106. Wylie, C. (1984) Median Lethal Dose (LD50) in
Rats. DuPont Haskell Laboratory Report HLR 433-83.
MRID
40042107 Gargus, J. and J. Sutherland (1983) Acute Skin
Absorption LD50 Test on Rabbits. Haskell Laboratory Report
HLO 288-83.
MRID
40357501 Dashiell, 0. (1982) INH-6573 Eye hitation Test
in Rabbits. DuPont Haskell Laboratory Report HLR 582-82.
To be submitted with registration application. Clouzeau,
J. (1991) Acute Dermal Imtation in the Rabbit. 7443 TAL.
/-
MRID
40357502 Dashiell, 0. (1982) IN H6573 Primary Skin Imtation
and Sensitization Test on Guinea Pigs. DuPont Haskell
Laboratory Report HLR 626-82.
SUBCHRONIC
TOXICITY
MRIDs 00072421 and 00161400. Pastoor,
T. (1983) Ninety-day Feeding and One-Generation Reproduction
Study in Rats with Silane. DuPont Haskell Laboratory Report
483-83
MRID 400421 11. Pastoor, T. (1983) Four-week
Range Finding and Ninety-Day Feeding in Mice with Silane,
[Bis(4-Fluorophenyl)](methyl) (1H-1,2,4-biazol-I-methyl)
(IN H-6573). DuPont Haskell Laboratory Report HLR 341-83.
MRID 41514901. Keller, D. (1990) Subchronic
Oral Toxicity: 90-Day Study with DPX- H6573-193 Feeding
Study in Mice. DuPont Haskell Laboratory Report HLR 60-90.
MRID 00161 168. Rickard, R. (1983) Three-month
Feeding Study in Dogs with Silane (INH- 6573). DuPont
Haskell Laboratory Report HLR 461-83.
MRID 400421 19. Sarver, J. (1986) Twenty-one
Day Dose Dermal Toxicity Study with INH- 6573-82 in Rabbits.
DuPont Haskell Laboratory Report HLR 744-86.
LONGTERM
TOXICITY AND CARCINOGENICITY
MRID 00148511. Pastoor, T. (1984) Long-term
Feeding and Two-generation, Four-litter Reproduction Study
in Rats with Silane, [Bis(4-Fluorophenyl)](methyl) (1H-1,2,4-Triazol-I-
ylmethy1)-(INH-6573). DuPont Haskell Laboratory Report
HLR 281-84 MRID 42613202. Keller, D. (1992) Oncogenicity
Study with DPX-H6573-194 (Flusilazole) 2-year Feeding
in Rats. DuPont Haskell Laboratory Report HLR 527-92.
MRID 400421 14. Brock, W. (1985) Long-term
Feeding Study in Mice with INH-6573. DuPont Haskell Laboratory
Report HLR 278-85.
MRID 42613201. Keller, D. (1992) Oncogenicity
Study with DPX-H6573-193 (Flusilazole) 18-month Feeding
in Mice. DuPont Haskell Laboratory Report HLR 35-92.
MRID 400421 13 O'Neal, F. (1985) One-year
Feeding Study in Dogs with IN H-6573. DuPont r Haskell
Laboratory Report HLR 461-85.
REPRODUCTIVE
AND DEVELOPMENTAL TOXICITY
MRIDs 00072421,00161400. Pastoor, T. (1983)
Ninety-day Feeding and One-generation Reproduction Study
in Rats with Silane. DuPont Haskell Laboratory Report
HLR 483-83.
MRID 0014851 1 Pastoor, T. (1984) Long-term
Feeding and Two-generation, Four-litter Reproduction Study
in Rats with Silane, [Bis(4-Fluorophenyl)](methyl) (1H-1,2,4-Triazol-1-
ylmethy1)-(INH-6573. DuPont Haskell Laboratory Report
HLR 281-84
MRID 41684601. Mullin, L. (1990) Reproductive
and Fertility Effects with Flusilazole Multigeneration
Reproduction Study in Rats. DuPont Haskell Laboratory
Report HLR 424- 90.
MRID 00072999. Alvarez, L. (1984). Developmental
Toxicity Study in Rats Given INH- 6573-66 in the Diet
on Days 7-16 of Gestation. DuPont Haskell Laboratory Report
HLR 431- 84.
MRID 00161 169. Lamontia, C. (1983) Embryo-fetal
Toxicity and Teratogenicity Study of INH-6573-39 by Gavage
in the Rat. DuPont Haskell Laboratory Report HLR 444-83.
MRID 001 61 170. Lamontia, C. (1 984)
Embryo-Fetal Toxicity and Teratogenicity Study of INH-6573-39
by Gavage in the Rat. DuPont Haskell Laboratory Report
HLR 142-84.
MRID 40640704. Alvarez, L. (1985) INH-6573:
Prenatal and Postnatal Toxicity Study in Rats Dosed by
Gavage on Days 7-16 of Gestation. DuPont Haskell Laboratory
Report 654-85.
MRID 45042601. Munley, S. (2000) Flusilazole
Technical: Developmental Toxicity Study in Rats. DuPont-2287.
MRID 44594201. Schardein, J. (1998) A Dermal Prenatal
Development Toxicity Study of Flusilazole in Rats. DuPont
Haskell Laboratory Report HLO-1998-01504 Revised.
MRID 00154930. Alvarez, L. (1985) INH-6573
Developmental Toxicity Study in Rabbits Treated by Diet
on Days 7-19 of Gestation. DuPont Haskell Laboratory Report
337-85.
MRID 00148512. Solomon, H. (1984) Developmental
Toxicity Study in Rabbits Given INH- 6573 by Gavage on
Days 7-19 of Gestation: DuPont Haskell Laboratory Report
HLR 333-84.
MRID 00154929. Zellers, J. (1985) INH-6573.
Developmental Toxicity Study (Supplemental) in Rabbits
Dosed by Gavage on Days 7-19 of Gestation. DuPont Haskell
Laboratory Report HLR 669-85. ,p
To be submitted with registration application.
Alvarez, L. (1990) Teratogenicity Study of DPX-H6573-66
in Rabbits. DuPont Haskell Laboratory Report HLR 216-90.
GENOTOXICITY
MRID
00161 171. Donovan, S. (1982) Mutagenicity Evaluation
in Salmonella typhimurium [of Silane]: MR No. 4581-047.
DuPont Haskell Laboratory Report No. 61 1-82.
To be submitted with registration application.
Vlachos, D. (1989). Evaluation of INH-6573-82 in the In
Vitro Assay for Chromosome Aberrations in Human Lymphocytes.
DuPont Haskell Laboratory Report HLR 745-86 Revision I.
MRID 00161 172. McCooey, K. (1983) CHOiHGPRT
Assay for Gene Mutation [using Silane]. DuPont Haskell
Laboratory Report HLR 449-83.
MRID 400421 17. Chromey, N. (1983) Unscheduled
DNA SynthesisRat Hepatocytes in vitro. DuPont Haskell
Laboratory Report HLR 209-83
To be submitted with registration application.
Vlachos, D. (1989). Mouse Bone Marrow Micronucleus Assay
of INH-6573-69. DuPont Haskell Laboratory Report HLO 437-84
Revision 1.
MRID 00161 173. Cortina, T. (1983) In
vivo Bone Marrow Chromosome Study in Rats with H# 14,728:
Final Report: HLA Project No. 201-614.
METABOLISM
MRID 400421 15. Anderson, J. et al., (1986)
Metabolism of [Carbon 141-DPX-H6573 in Rats. DuPont AMR
196-128.
MECHANISTIC
STUDIES
MRID 42613204. Keller, D. (1992) Mechanisms
of Toxicity: 90-day Feeding Study in Rats with DPX-H6573-194
(Flusilazole): Revision No. 1. DuPont Haskell Laboratory
Report HLR 628-92.
To be submitted with registration application.
Cook, J. (1993) Mechanisms of Rat Leydig Cell Tumor Induction
by DPX-H6573-193 (Flusilazole) (Revision 1) Supplement
to Oncogenicity Study with DPX-H6573-194 (Flusilazole)
Two-Year Feeding Study in Rats (HLR 527-92). DuPont Haskell
Laboratory Report HLR 410-93.
WORKER
EXPOSURE
To be submitted with registration application.
Old, J. et al. (2002) Monitoring Exposure of I- Workers
During Mixing, Loading, and Application Using Flusilazole
25EW Fungicide for Control of Cereal Diseases in the UK:
Field Phase and Routine Analysis. Inveresk Report No.
20690 DuPont-3899.
FOLIAR
DISSIPATION
To
be submitted with registration application. Smyser, B.
(1992) Dissipation of Flusilazole from Wheat Forage. DuPont
Report AMR 1855-90.
DuPont
states: The following reports have not been submitted
to US EPA, but are available for review upon request.
Brodsky, J. 1991. Determination of Residues
of Flusilazole (DPX-H6573) in Soybeans by GC-MS following
Treatment with "Punch CS" (Season 1990 - France).
Battelle-Institut E.V. Frankfurt, Germany. BE-A-I I-91-01-BF.
Unpublished.
DuPont Brazil Study Numbers: RBR-04-276,
RBR-04-277, RBR-04-278; Study Director: AndrC Luis Moraes.
2004. Unpublished.
San Juan, M., Morre, J. 2004. Magnitude
of Residues of DPX-H6573 (Flusilazole) in Cultivars of
Soybean (Glycine mat L. Merr.) for the Registration of
the Product FUSION@ (Fungicide) (Flusilazole 12.5% + Carbendazim
25% SC) Trials canied out in the Argentine Republic Season
2003. DuPont Argentina Study Number: 005-2004. Unpublished.
Garbers, H.V. 2003. Determination of Flusilazole
and Carbendazim Residues in Soya. SABS, Pretoria, South
Africa. Report No. 721412126256iW393. . Unpublished. |
From
Dart Special at Toxnet
Toxicol
Lett 2003 Sep ;144 (Suppl 1 ):S107
Mechanisms
Involved In Triazole-Induced Teratogenesis: In Vitro Study.
Massa V, Broccia ML, Di Renzo F, Menegola
E, Giavini E
University
of Milan- Department of Biology, 20133 Milan, Italy.
Triazole-derivatives are antimycotic compounds used in agricolture
as well as in clinical and veterinary therapy. Literature data,
confirmed by results obtained in our previous studies, showed
severe alterations at the level of the branchial apparatus after
triazoles in vitro exposure (hypoplasia of I and II branchial
arches, fusion between I and II branchial arches). Our previous
works on Fluconazole (FLUCO) showed that FLUCO exposure is able
to alter the morphogenesis of the branchial apparatus modifying
the rhomboencephalic neural crest cells (NCC) migration in rat
embryos cultured in vitro. The hindbrain segmentation was also
altered by FLUCO. The aim of the present work was to extend
the study of the mechanisms to other molecules of this class
to verify if different molecules of the same family have the
same target. For this purpose 9.5 d.p.c. old rat embryos were
exposed in vitro to Flusilazole (25
uM), Triadimefon (250 uM) and Triadimenol (125 uM). The morphology
of the embryos was analysed after 48 hours of culture. Some
embryos cultured for 60 hours were immunostained using antibodies
anti-160 kDa neurofilament in order to evaluate the cranial
nerve structures. The localisation and distribution of NCC was
evaluated after 24, 30 and 48 hours of culture, using the specific
immunostaining of CRAB proteins. The expression and localisation
of Hox bl and Krox 20 proteins (used as markers for the study
of the correct hindbrain segmentation) was evaluated using whole
mount immunostaining after 24 hours of culture. The obtained
results showed very similar effects after exposure to Flusilazole,
Triadimefon and Triadimenol: abnormalities
at the level of the branchial apparatus; disorganisation and
fusions at the level of the cranial nerves; abnormalities in
the migration of NCC, not able to form 3 distinct migration
stripes from the rhomboencephalon to the branchial apparatus;
alteration of the hindbrain segmentation, with reduced and scattered
immunolocalised stripes. The collected data suggest a
common target for all the examined molecules: the observed branchial
abnormalities are due to anomalous NCC migration related to
an incorrect organisation and specification of the rhomboencephalon.
http://www.ncbi.nlm.nih.gov:80/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=11489598&dopt=Abstract
Reprod Toxicol
2001 Jul-Aug;15(4):421-7
Antifungal
triazoles induce malformations in vitro.
Menegola E, Broccia ML, Di Renzo
F, Giavini E.
Department of Biology, University of Milan, Italy.
Triazole-derivatives are antimycotics used in agriculture as
well as in clinical and veterinary therapy. The aim of the present
work is the in vitro comparative study of the teratogenic activity
of triazole (the parental compound), flusilazole
(an agricultural triazole mono-derivative fungicide), and fluconazole
(a clinically used bis-triazole derivative). Rat embryos, 9.5
days old (1 to 3 somites) were exposed in vitro to triazole
500 to 5000 microM, flusilazole 3.125 to 250 microM, or fluconazole
62.5 to 500 microM. After 48 h in culture, the embryos were
morphologically examined and processed for histologic and biochemical
analysis. Flusilazole and fluconazole showed similar teratogenic
effects (abnormalities at the branchial
apparatus level and cell death at the level of the branchial
mesenchyme) at concentration levels of 6.25 microM and
higher for flusilazole and of 125 microM and higher for fluconazole.
By contrast, only slight developmental retardation and blood
discoloration were observed at the highest concentrations of
triazole, suggesting no teratogenic activity for the triazole
group.
PMID: 11489598 [PubMed - indexed for MEDLINE]
http://www.ncbi.nlm.nih.gov:80/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=10794648&dopt=Abstract
J Agric
Food Chem 1999 Jun;47(6):2439-46
Metabolism
of [(14)C]flusilazole in the goat.
Anderson JJ, Shalaby LM, Berg
DS.
Experimental Station, DuPont Agricultural
Products, Wilmington, Delaware 19880-0402, USA.
[Phenyl(U)-(14)C] and [triazole(3)-(14)C]flusilazole ([(bis
4-fluorophenyl)]methyl(1H-1,2,4-triazole-1-ylmethyl)silane;
I) were extensively metabolized when fed to lactating goats
(Capra hircus). The primary metabolites identified in goat tissues
and milk were bis(4-fluorophenyl)(methyl)silanol (II) and 1H-1,2,4-triazole
(III). Concentrations of total radiolabeled residues in the
milk ranged from 0.09 to 0.74 microg/mL. Concentrations of radiolabeled
residues found in tissues when the [(14)C] label was in the
phenyl or triazole position, respectively, were 13.5 and 3.54
microg/g (liver), 8.74 and 0.75 microg/g (kidney), 0.41 and
0.52 microg/g (leg muscle), and 4.07 and 0.94 microg/g (back
fat). Urine contained an additional major metabolite identified
as [bis(4-fluorophenyl)](methyl)silylmethanol (IV) and its glucuronic
acid conjugate (V). With either labeled form of flusilazole,
the majority of the recovered radiolabel was excreted in urine
or feces.
PMID: 10794648 [PubMed - indexed for MEDLINE]
From
Toxline at Toxnet
Pesticide
residues in food - 1995. Toxicology
evaluations (1996) pp 157-79
Flusilazole
Authors:
FAO and WHO working groups
Levels that
cause no toxic effect. Mouse: 25 ppm, equal to 3.4 mg/kg bw
per day (18-month study of toxicity and carcinogenicity). Rat:
10 ppm, equal to 0.4 mg/kg bw per day (two-year study of toxicity
and carcinogenicity); 5 ppm, equal to 0.34 mg/kg bw per day
(maternal toxicity in a two-generation study of reproductive
toxicity); 50 ppm, equal to 4 mg/kg bw per day (two-generation
study of reproductive toxicity); 4.6 mg/kg bw per day (embryo-
or fetotoxicity in a study of developmental toxicity); 100 mg/kg
bw per day (teratogenicity in a study of developmental toxicity).
Rabbit: 12 mg/kg bw per day (maternal and embryo- or fetal toxicity
in a study of developmental toxicity); 15 mg/kg bw per day (teratogenicity
in a study of developmental toxicity). Dog: 5ppm, equal to 0.14
mg/kg bw per day (one-year study of toxicity). Estimate of acceptable
daily intake for humans. 0-0.001 mg/kg bw.
From Dart Special at Toxnet
Chemically Induced Birth Defects 1993;2:675-721
Pesticides.
Schardein JL
International Research and Development Corporation, Mattawan,
MI.
Medical Subject Headings (MeSH):
Pregnancy
Animal
Human
Female
Pesticides/*TOXICITY
*Abnormalities, Drug-Induced
2,4,5-Trichlorophenoxyacetic Acid/TOXICITY
Insecticides/TOXICITY
Substance (CAS Registry Number): [Too
many to list]
Sodium fluoroacetate (62-74-8)
[Note: the following organofluorines
were included]
Diflubenzuron
(35367-38-5)
Ethalfluralin
(55283-68-6)
Flusilazole
(85509-19-9)
Gliftor (8065-71-2) - [Synonym: 1-Chloro-3-fluoro-2-propanol
mixt. with 1,3-difluoro-2-propanol]
N-Methyl-N- 1-naphthyl fluoroacetamide [Nissol] (5903-13-9)
Sarin [Synonym: (+-)-Isopropyl methylphosphonofluoridate] (107-44-8)
Sodium
fluoroacetate (62-74-8)
Sodium
hexafluorosilicate [also known as Sodium fluorosilicate]
(16893-85-9)
Soman [Synonym: 1,2,2-Trimethylpropyl methylphosphonofluoridate]
(96-64-0)
Sulfuryl
fluoride (2699-79-8)
Trifluralin
(1582-09-8)
From
Toxline at Toxnet
FAO Plant
production and protection paper Vol:100/2 (1988)
pp 117-39
Flusilazole
Authors:
FAO and WHO working groups
Level causing
no toxicological effect Mouse: 25 ppm in the diet, equal to
3.4 mg/kg bw/day Rat: 10 ppm in the diet, equal to 0.45 mg/kg
bw/day Dog: 5 ppm in the diet, equal to 0.14 mg/kg bw/day Estimate
of acceptable daily intake for humans 0-0.001 mg/kg bw. Studies
which will provide information valuable in the continued evaluation
of the compound 1. Observations in humans. 2. Toxicity of the
biotransformation products.
Helpful
background reading:
2002 - Zerulla
M, Lþnge R, Steger-Hartmann T, Panter G, Hutchinson T,and Dietrich
DR.
Morphological sex reversal upon short-term exposure to endocrine
modulators in juvenile fathead minnow (Pimephales promelas).
Toxicology Letters 131: 51-63.
2001 - Blzquez
M, Felip A, Zanuy S, Carrillo M, Piferrer F.
Critical period of androgen-inducible sex differentiation in
a teleost fish, the European sea bass.
Journal of Fish Biology 58: 342-358.
1998 - Nagel,
R. and Isberner, K.
Testing of chemicals with fish Ð a critical evaluation of tests
with special regard to zebrafish.
In: Braunbeck,T., Hinton, D.E. and Streit, B.: Fish Ecotoxicology.
Basel, pp337-352.
1997 - Peter,
H. and Heger, W.
Long-term effects of chemicals in aquatic ecosystems.
In: Schrmann, G. and Markert, B. (Eds.) Ecotoxicology. New
York and heidelberg, pp 571-586.
1997 - Trant
JM, Lehrter J, Gregory T, Nunez S, and Wunder J.
Expression of cytochrome P450 aromatase in the channel catfish,
Ictalurus punctatus.
J. Steroid Biochem. Molec. Biol. 61: 393-397.
1977 - Macek,
K.J. and Sleight, B.H.
Utility of toxicity tests with embryos and fry of fish in evaluating
hazard associated with the chronic toxicity of chemicals to
fishes.
Eds. F.L. Meyer and J.L. Hamelink, pp.137 Ð 146. ASTM STP 634.
Philadelphia: ASTM 1977.
1977 - Mc
Kim, J.M.
Evaluation of tests with early life stages of fish for predicting
long-term toxicity.
J. Fish Res Bd Can. 34: 1148 Ð 1154.
Return
to Flusilazole
Index Page
|