|
Return to
Index Page
Adverse
Effects
ACTIVITY:
Herbicide
(pyridazinone)
CAS Name
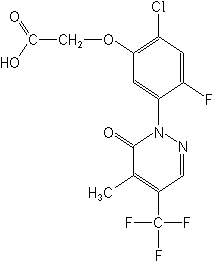
for Flufenpyr:
[2-chloro-4-fluoro-5-[5-methyl-6-oxo-4-(trifluoromethyl)-1(6H)-pyridazinyl]phenoxy]acetic
acid
NOTES:
This compound is normally used as a salt or an ester, the identity
of which should be stated, e.g. flufenpyr-ethyl [188489-07-8].
Structure
for Flufenpyr:
| |
| Published
Date |
Docket
Identification Number |
Details |
|
Sept
19, 2003
|
OPP-2003-0166
|
Valent
- Flufenpyr-ethyl
- Pesticide
tolerances. FINAL RULE.
-- No
tolerances have been previously established for the residues
and the combined residues of flufenpyr-ethyl, in or on raw agricultural
commodities.
-- C. Conditions
Confirmatory storage stability data for the metabolite S-3153
acid- 4-OH in field corn forage and stover and an
enforcement
method for measuring both parent and metabolite are required.
| Tolerances
are established for residues of the herbicide, flufenpyr-ethyl;
acetic acid |
| Commodity |
Parts
per million |
| Corn,
field, grain
Soybean, seed
Sugarcane, cane |
0.01 |
| Tolerances
are established for residues of the herbicide flufenpyr-ethyl;
acetic acid, and its metabolite,
S-3153 acid-4-OH; [2-chloro-4-hydroxy-5-[5-methyl-6- oxo-4-
(trifluoromethyl)-1-(6H)-pyridazinyl]-phenoxy]-acetic
acid, free and conjugated, in or on the following commodities: |
Corn,
field, forage
Corn, field, stover |
0.05 |
| Some
excerpts from Table 1.--Subchronic, Chronic, and Other
Toxicity |
Study
type
[Guideline No.] |
Results |
| Note:
no results were identified from rat studies |
90-Day oral toxicity rodents
(mouse) - [870.3100] |
NOAEL
= 395 mg/kg/day (M) LOAEL = 908 mg/kg/ day, based
on increased absolute and relative liver weights and increased
incidence of hepatic centrilobular vacuolation in male
mice |
28-Day
oral toxicity rodents
(mouse) - [870.3100] |
NOAEL
= 448/629 mg/kg/ day M/F LOAEL = 1,009/1,213 M/ F mg/kg/day,
based on increased incidence of
hepatic centrilobular vacuolation |
90-Day oral toxicity in non/ rodents
(dog) - [870.3150] |
NOAEL
= 300 mg/kg/day M/F LOAEL = 1,000 M/F mg/ kg/day, based
on decreased body weight gains, food consumption, and
food efficiency and increased incidence of vomiting |
Prenatal
developmental in nonrodents
(rabbit) - [870.3700] |
Maternal
NOAEL = 100 mg/kg/day LOAEL = 300 mg/kg/day, based
on increased maternal mortality, clinical signs, decreased
food consumption and necropsy findings Developmental
NOAEL = 1,000 mg/kg/ day LOAEL not identified |
Prenatal developmental in nonrodents
(rabbit) - [870.3700] |
Maternal
NOAEL = 100 mg/kg/day LOAEL = 200 mg/kg/day,
based on increased mortality Developmental NOAEL
= 1,000 mg/kg/ day HDT LOAEL not identified |
Carcinogenicity
rodents
(mouse) - [870.4200] |
NOAEL
= 39.9 - 43.7 mg/kg/day M/F LOAEL = 401.8 - 447.9 mg/kg/day
M/F, based on liver toxicity in
both sexes and mild anemia in males No evidence of carcinogenicity |
| Some
excerpts from Table 2.-Summary of Toxicological Dose and
Endpoints for Flufenpyr-ethyl for Use in Human Health
Risk Assessment1 |
| Note:
no results were identified from rat studies |
| --Chronic
dietary (all populations)
-- Short-term inhalation (1-30 days)
--Intermediate-term inhalation (1-6 months)
-- Long-term inhalation (>6 months) |
Carcinogenicity
study - mice.
NOAEL = 40 mg/kg/day
LOAEL = 401.8 mg/kg/day based on
liver toxicity (hepatocyte necrosis) in both sexes and
mild anemia in males |
| Short-term
Incidental oral (1-30 days) and (1-6 months) |
Developmental toxicity study - rabbit
NOAEL = 100 mg/kg/day LOAEL = 300 mg/kg/day, based
on clinical signs, decreased food consumption and necropsy
findings |
|
| June
25, 2003 |
OPP-2003-0166 |
Valent.
Flufenpyr-ethyl. Pesticide
Tolerance petition; to
establish tolerances for residues of the herbicide chemical
flufenpyr-ethyl in or on the raw agricultural commodities corn,
field, grain; soybean, seed; and sugarcane, cane at 0.01 ppm
and for the combined residues of the herbicide
chemical flufenpyr-ethyl, and
its metabolite, S-3153-acid-4-OH, [2-chloro-4-hydroxy-5-(5-methyl-6-oxo-4-
trifluoromethyl-1,6-dihydropyridazin-1-yl)phenoxy]acetic acid,
free and conjugated, in or on the raw agricultural commodities
corn, field, forage and corn, field, stover
at 0.05 ppm.
Note from FAN: In animal studies the males are more susceptible.
-- The kidney and liver appear to be the target organs of flufenpyr-ethyl.
EPA has not had the opportunity to review
the toxicity studies on flufenpyr-ethyl and has not established
toxic endpoints.
-- A second developmental toxicity study
was conducted to confirm the maternal NOAEL at dose levels of
0, 100, 200, 300 or 1,000 mg/kg/day during gestation. Again,
maternal mortality occurred, but at all
dose levels. Detailed examination
of these animals showed in the majority of cases the cause of
death to be test material aspiration into the lungs. The cause
of death for several animals at the high dose could not be determined.
Their deaths were therefore attributed to systemic toxicity.
-- Reproduction. In the rat reproduction study, flufenpyr-ethyl
technical was administered in the diet at levels of 0, 200,
2,000, and 20,000 ppm for 2-generations.
Parental toxicity was observed at all dose levels... Parental
toxicity was exhibited by dose-related microscopic changes
in the kidney in high dose F0
animals, in all treated F1
males, and in high dose F1
females. There were also 2 high dose F1 males
that died possibly as a result of treatment. Midzonal cytoplasmic
vacuolation of the hepatocytes was also observed in the liver
of all groups of treated animals in both generations.
-- A second 1-generation reproduction
study was performed to establish a clear NOAEL for adult kidney
lesions using the dose levels of 20, 50 and 100 ppm. The results
of the study indicate that the NOAEL for histological
changes in the kidneys of F1 male rats was
100 ppm.
-- Rats. Pure (99.4%) flufenpyr-ethyl
was tested in rats at dose levels of 0, 600, 2,000, 6,000, and
20,000 ppm in the diet for 13 weeks. Effects observed included
urinary incontinence, increased food and water consumption,
slight hematological and blood biochemistry
changes, decreased spleen weights, an increase in the incidence
and severity of basophilic tubules of the kidneys and slight
to mild diffusely distributed vacuolation in the liver.
Based on these results, the NOAEL was 2,000 ppm (134.2 mg/kg/day)
for the males and 20,000
ppm (1,509.6 mg/kg/day) for the females.
-- In an additional study, flufenpyr-ethyl technical was tested
in rats at dose levels of 0, 1,000, 10,000, and 20,000 ppm in
the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, and mild urinalysis, hematological
and blood biochemistry changes. Thymus weights were slightly
increased. Diffusely distributed hepatic vacuolation was seen
in the high dose males. Based on these findings, the NOAEL was
10,000 ppm (595.2 mg/kg/ day) in the males
and 20,000 ppm (1,377.5 mg/kg/day) in the females. |
| Nov
13, 2002 |
OPP-2002-
0279 |
Valent.
Flufenpyr-ethyl.
Pesticide Products; Registration Applications.
1. File
symbol: 59639-RNO.Product name:
S-3153 Flufenpyr-ethyl
Technical. Product type: Herbicide. Active ingredient:
Flufenpyr-ethyl, ethyl [2-chloro-4- fluoro-5-(5-methyl-6-oxo-4-trifluoromethyl-1,6-dihydropyridazin-1-
yl)phenoxy]acetate at 98.0%.
Proposed classification/Use: None. For
formulation into herbicide products to control postemergence
broadleaf weed species in field corn, forage; field corn,
grain; field corn, stover; soybean, seed; sugarcane.
2. File
symbol: 59639-RRN. Product name: S-3153
WDG Herbicide. Product type: Herbicide. Active
ingredient: Flufenpyr-ethyl at 57.6%.
Proposed classification/Use: None. For
manufacturing use of end-use products to be used to control
postemergence broadleaf weed species in field corn, soybeans
and sugarcane.
3. File
symbol: 59639-RRR. Product name: S-3153 Atrazine WDG.
Product type: Herbicide. Active ingredient: Flufenpyr-ethyl
75.0%.
Proposed classification/Use: None. For
manufacturing use of end-use products to be used to control
postemergence broadleaf weed species in field corn and sugarcane.
|
|

