|
ACTIVITY:
Fungicide
(azole)
Synonyms for
Triflumizole [BSI:ISO] - CAS No. 68694-11-1
• (E)-4-Chloro-alpha,alpha,alpha-trifluoro-N-(1-imidazol-1-yl-2-propoxyethylidene)-o-toluidine
• 1-(1-((4-Chloro-2-(trifluoromethyl)phenyl)imino)-2-propoxyethyl)-1H-imidazole
• 4-Chloro-alpha,alpha,alpha-trifluoro-N-(1-imidazol-1-yl-2-propoxyethylidene)-o-toluidine
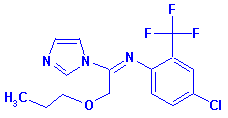
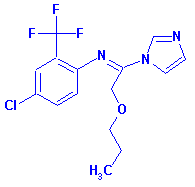
| Structure
for CAS No. 99387-89-0 |
Structure
for CAS No. 68694-11-1 |
 |
 |
Report
available from
The National Technical Information Service
(NTIS)
Order from NTIS by: phone at 1-800-553-NTIS (U.S. customers);
(703)605-6000 (other countries); fax at (703)605-6900; and
email at orders@ntis.gov. NTIS is located at 5285 Port Royal
Road, Springfield, VA, 22161, USA. |
| Order
No. |
Title |
Abstract |
NTIS/PB92-126978
9p
|
1991
- Pesticide Fact Sheet Number 228: Triflumizole.
Environmental
Protection Agency, Washington, DC. Office of Pesticides
and Toxic Substances. |
The
document contains up-to-date chemical information, including
a summary of the Agency's regulatory position and rationale,
on a specific pesticide or group of pesticides. A Fact
Sheet is issued when the registration of a new chemical
has taken place as in the case with Triflumizole. |
CAS
No. 68694-11-1
From
Toxline at Toxnet
J
PESTIC SCI; 16 (2). 1991.
301-314.
Development
of a new fungicide, triflumizole.
NAKATA A, HASHIMOTO S, IKURA K,
KATSUURA K
Odawara
Res. Cent., Nippon Soda Co. Ltd., Takada, Odawara 250-02,
Jpn.
BIOSIS COPYRIGHT: BIOL ABS. Triflumizole (E-4-Chloro-alpha,alpha,alpha-trifluoro-N-(1-imidazol-1-yl-2-propoxyethylidene)-o-toluidine)
is a novel systemic fungicide discovered
and developed by Nippon Soda Co., Ltd. We found
a new synthetic method of imidazole derivatives and
we used this method for the developing of new pesticide.
The synthesized imidazole containing compound, N-(1-imidazol-1-ylalkoxyalkylidene)anilines
showed relatively strong and broad fungicidal activity
accompanied with plant growth regulating effect. T highest
fungicidal activity was obtained by phenyl substitution
with halogen, methyl or trifluoromethyl in the 2-position
and halogen in the 4-position, respectively. The introduction
of hetero atom, 0, to alkyl chain reduced the effect
on plant growth remarkably without reducing fungicidal
efficacy. As the results, a compound was selected and
is now marketed as a common name triflumizole. Triflumizole
showed a wide range of antifungal spectrum in vitro
including fungicidal activ [abstract truncated]
|
No CAS No. Cited
Crop Protection; Volume 23, Issue 5
, May 2004, Pages 371-378
Mechanism for the differential
toxicity of neonicotinoid insecticides in the honey
bee, Apis mellifera
Takao Iwasa, Naoki Motoyama,
John T. Ambrose and R.M.R. Michael Roe
Department of Entomology, Dearstyne
Entomology Building, Campus Box 7647, North Carolina
State University, Raleigh, NC 27695-7647, USA
Laboratory bioassays were conducted to determine the
contact honey bee toxicity of commercial and candidate
neonicotinoid insecticides. The nitro-substituted compounds
were the most toxic to the honey bee in our laboratory
studies with LD50 values of 18 ng/bee for imidacloprid,
22 ng for clothianidin, 30 ng for thiamethoxam,
75 ng for dinotefuran and 138 ng for nitenpyram.
The cyano-substituted neonicotinoids exhibited a much
lower toxicity with LD50 values for acetamiprid and
thiacloprid of 7.1 and 14.6 g/bee, respectively.
Piperonyl butoxide, triflumizole
and propiconazole increased
honey bee toxicity of acetamiprid 6.0-, 244-
and 105-fold and thiacloprid 154-, 1,141-
and 559-fold, respectively, but had a minimal
effect on imidacloprid (1.70, 1.85 and 1.52-fold, respectively).
The acetamiprid metabolites, N-demethyl acetamiprid,
6-chloro-3-pyridylmethanol and 6-chloro-nicotinic acid
when applied topically, produced no mortality at 50 g/bee.
These results suggest that P450s are an important mechanism
for acetamiprid and thiacloprid detoxification and their
low toxicity to honey bees. When honey bees were placed
in cages in forced contact with alfalfa treated with
acetamiprid and the synergist, triflumizole, in combination
at their maximum recommended application rates, no mortality
was detected above that of the control.
|
No CAS No. Cited
From Toxline at
Toxnet
MEDEDELINGEN FACULTEIT LANDBOUWKUNDIGE EN TOEGEPASTE BIOLOGISCHE
WETENSCHAPPEN UNIVERSITEIT GENT; 62 (3B). 1997.
1103-1107.
Screening hormonal effects of fungicides
with a Ficus benjamina L. chimera.
WERBROUCK S PO, JUMLI, DE ROECK K, EECKHAUT
T GR, DEBERGH PC
Lab. Hortic., Fac. Agric. Appl. Biol. Sci., Univ. Gent, Coupure
Links 653, B-9000 Gent, Belgium.
BIOSIS COPYRIGHT: BIOL ABS. A Ficus benjamina chimera with an
albino L2- and L3-layer and a genetic green L1-layer can be
used to detect interactions between pesticides and plant hormones.
When this chimera was multiplied in vitro, the leaves were yellow
green because the epidermis (L1) contained only few chloroplasts,
Adding fungicides such as prochloraz,
imazalil, triflumizole, bittertanol,
dodemorf and carbendazim to this medium
disturbed the integrity of the cell layers in the meristems.
Dark green spots on newly developed leaves indicated that cells
from the L1-layer intruded the L2 layer. These cells look green
because they develop like normal L2-cells with chloroplasts.
For prochloraz and possibly also for the other ergosterole biosynthesis
inhibiting fungicides an inhibition of the GA biosynthesis might
be an explanation. The effects of carbendazim could be explained
by its known cytokinin-like properties.
CAS NO. 68694-11-1 [several
other CAS Nos. were listed]
ARCHIVES OF ENVIRONMENTAL CONTAMINATION AND TOXICOLOGY; 28
(1). 1995. 1-7.
Effects of pesticides on isolated rat
hepatocytes, mitochondria, and microsomes II.
YAMANO T, MORITA S
The hepatotoxic effects of 22 pesticides were studied. Hepatocytes,
mitochondria, and microsomes were prepared from Sprague-Dawley-rats
and incubated with the test substances in-vitro. The nonprotein
sulfhydryl (NPSH) contents of hepatocytes were decreased by
12 of the pesticides, most markedly by thalonil (1897-45-6)
and ziram (137-30-4). The NPSH depleting compounds copper-terephthalate
(34262-89-0) and chlorothalonil (1897-45-6) also reacted with
glutathione. Hepatic lipid peroxidation was induced by chlorothalonil,
pretilachlor (51218-49-6), ethoprofos (13194-48-4), and metribuzin
(21087-64-9). Hepatic mitochondrial respiration
was affected by alpha-endosulfan (959-98-8), ethoprofos,
flutolanil (66332-96-5), mepronil (55814-41-0), inabenfide (82211-24-3),
trichlamide (70193-21-4), triflumizole
(68694-11-1), tralomethrin (66841-25-6), bitertanol (55179-31-2),
pendimethalin (40487-42-1), and sethoxydim (74051-80-2) while
microsomal lipid peroxidation was inhibited by trichlamide,
copper-terephthalate, and ziram, and stimulated by etrimfos
(38260-54-7), glufosinate (51276-47-2), and metribuzin. Studies
examining mechanisms associated with the cytotoxicity of the
three most toxic compounds suggested that a decrease in cellular
sulfhydryls was related to the toxicity of chlorothalonil and
ziram, while depletion of ATP was seen prior to cell death following
incubation with trichlamide.
CAS NO. 68694-11-1
[Two other CAS Nos. were listed]
BURDITT, A. K. JR. (ED.). ARTHROPOD MANAGEMENT TESTS, VOL.
20. III+399P. ENTOMOLOGICAL SOCIETY OF AMERICA: LANHAM, MARYLAND,
USA. ISBN 0-938522-53-1.; 0 (0). 1995.
331.
LABORATORY BIOASSAY OF SELECTED BT INSECTICIDES
AGAINST TOBACCO BUDWORM 1994
WHITE CA, LEONARD BR, BURRIS G, GRAVES
JB
BIOSIS COPYRIGHT: BIOL ABS. RRM BOOK CHAPTER GOSSYPIUM-HIRSUTUM
HELIOTHIS-VIRESCENS CONDOR DESIGN DIPEL JAVELIN MVP RAPTOR LARVIN
INSECTICIDE PESTICIDE EVALUATION ARTHROPOD MANAGEMENT TEST
CAS Registry Numbers:
100920-69-2 - 3-Pyridinecarboxamide, N-(2,4-difluorophenyl)-2-(3-(trifluoromethyl)phenoxy)-,
mixt. with N,N-dimethyl-N'-(4-(1-
methylethyl) phenyl)urea
68694-11-1 - Triflumizole
59669-26-0 - Thiodicarb
CAS NO. 68694-11-1
[several other CAS Nos. were listed]
ANNUAL MEETING OF THE AMERICAN PHYTOPATHOLOGICAL ASSOCIATION,
PITTSBURGH, PENNSYLVANIA, USA, AUGUST 12-16, 1995. PHYTOPATHOLOGY;
85 (10). 1995. 1125.
IR-4 REGISTRATION OF MINOR USES OF FUNGICIDES
AND NEMATICIDES
THOMPSON DC, BIEHN WL, GUEST RT
BIOSIS COPYRIGHT: BIOL ABS. RRM MEETING ABSTRACT CROP INDUSTRY
AGRICHEMICAL RESIDUES POTENTIAL PROFITS
CAS Registry Numbers:
131341-86-1 - Fludioxonil
119611-00-6 - Fenethanil
119446-68-3 - Difenoconazole
107534-96-3 - Tebuconazole
68694-11-1 - Triflumizole
8018-01-7 - Mancozeb
1897-45-6 - Chlorothalonil
CAS NO. 68694-11-1
From Toxline at
Toxnet
JOURNAL OF PESTICIDE SCIENCE; 19 (1). 1994.
1-10.
Runoff and leaching of pesticides in
golf course.
ODANAKA Y, TANIGUCHI T, SHIMAMURA Y, IIJIMA
K, KOMA Y, TAKECHI T, MATANO
Mitsukaido Inst., Inst. Environmental Toxicol., Uchimoriya-cho,
Mitsukaido 303, JAP.
BIOSIS COPYRIGHT: BIOL ABS. An investigation of runoff property
of pesticides applied in a golf course was conducted by using
lysimeters and at actual golf courses. In lysimeter experiment,
a remarkable leaching was observed in only 5 pesticides (trichlorfon
(DEP), metalaxyl, isoprothiolane, flutolanil and iprodione)
with at least 100 mm of precipitation. The leaching was scarce
in other 12 pesticides (bensulide (SAP), isofenphos, fenitrothion
(MEP), fenarimol, triflumizole,
tetrachlorvinphos (CVMP), pyridaphenthion, oxine-copper, isoxathion,
prothiofos, tolclofosmethyl, pendimethalin) with sum of 440
mm of precipitation for 51 days. The leaching pattern of the
5 pesticides showed a single broad peak. There was a tendency
that pesticides with a high water solubility had a potential
to move downward. In putting green experiment, all the 3 pesticides
applied (isofenphos, flutolanil and metalaxyl) were detected
in leaching water with only 10 mm of precipitation. There was
no significant difference am [abstract truncated]
CAS NO. 68694-11-1
[several other CAS Nos. were listed]
PFLANZENSCHUTZ (VIENNA); 0 (2). 1992.
9-16.
TABLE OF LICENSED FUNGICIDES IN HORTICULTURE
FOR 1992
BEDLAN G
BIOSIS COPYRIGHT: BIOL ABS. RRM REVIEW CABBAGE
LETTUCE CELERY HORSERADISH CARROTS ONIONS BEANS PEAS CUCUMBERS
TOMATOES PEPPERS MUSHROOMS LIST
CAS. NO. 99387-89-0 [several
other CAS Nos. were listed]
FOURTH INTERNATIONAL WORKSHOP ON CHEMICAL, BIOLOGICAL AND ECOTOXICOLOGICAL
BEHAVIOUR OF PESTICIDES IN THE SOIL ENVIRONMENT, ROME, ITALY,
MAY 29-31, 1991. SCI TOTAL ENVIRON; 123-124 (0). 1992.
459-468.
ASSESSMENT OF THE MOVEMENT OF TRIAZOLE
FUNGICIDES BY SOIL THIN-LAYER CHROMATOGRAPHY
JAMET P, EUDELINE V
BIOSIS COPYRIGHT: BIOL ABS. RRM PENICILLIUM-EXPANSUM ENVIRONMENTAL
TOXICOLOGY SOIL BIOASSAY ANALYTICAL METHOD FRANCE
CAS NO. 68694-11-1
http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=pubmed&dopt=Abstract&list_uids=1885028
GIG SANIT; 1991 Mar;(3):83-4.
DETERMINATION OF TRIFLUMIZOLE AND ITS
TRANSFORMATION PRODUCTS IN AGRICULTURAL CROPS WATER AND SOIL
PIS'MENNAYA MV
No abstract available
PMID: 1885028 [PubMed - indexed for MEDLINE]
CAS NO. 68694-11-1
PFLANZENSCHUTZ (VIENNA); 0 (3). 1991.
10-16.
OVERVIEW OF FUNGICIDES PERMITTED IN
1991 FOR VEGETABLE CULTIVATION
BEDLAN G
BIOSIS COPYRIGHT: BIOL ABS. RRM ONIONS
LEGUMES MUSHROOMS FRUIT TUBERS ROOT VEGETABLES
CAS NO. 68694-11-1
PFLANZENSCHUTZ (VIENNA); 0 (SPEC. ISSUE 6). 1988.
6-12.
REVIEW OF ACTIVE SUBSTANCES AND APPROVED
COMMERCIAL PREPARATIONS AVAILABLE FOR PLANT PROTECTION IN FRUIT
GROWING
FISCHER-COLBRIE P
CAS NO. 68694-11-1
(approx. 23 other CAS Nos. were listed]
JPN PESTIC INF; 0 (50). 1987.
34-36.
1986 EVALUATION OF CANDIDATE PESTICIDES
B-III FUNGICIDES FRUIT TREES
KOIZUMI M
|