|
Return to
Index Page
Adverse Effects
Abstracts
ACTIVITY:
Fungicide
(azole)
Synonyms for
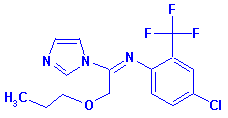
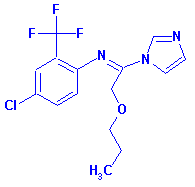
Triflumizole [BSI:ISO] - CAS No. 68694-11-1
• (E)-4-Chloro-alpha,alpha,alpha-trifluoro-N-(1-imidazol-1-yl-2-propoxyethylidene)-o-toluidine
• 1-(1-((4-Chloro-2-(trifluoromethyl)phenyl)imino)-2-propoxyethyl)-1H-imidazole
• 4-Chloro-alpha,alpha,alpha-trifluoro-N-(1-imidazol-1-yl-2-propoxyethylidene)-o-toluidine
| Structure
for CAS No. 99387-89-0 |
Structure
for CAS No. 68694-11-1 |
 |
 |
| |
| Date
Published |
Docket
Identification Number |
Details |
| June 27, 2007 |
EPA-HQ-OPP-2007-0312 |
IR-4.
Pesticide
Petition. PP 7E7183. Proposal to establish a tolerance
for residues of the fungicide triflumizole and its metabolites
containing the 4-chloro-2-trifluoromethylaniline moiety, calculated
as the parent compound in or on food commodity
leafy Brassica (subgroup
5B)
This subgroup includes
8 commodities.
broccoli raab • cabbage, chinese bok choy •
collards • kale • mizuna • mustard
greens • mustard spinach • rape greens
|
20.0 ppm |
The analytical method is suitable for analyzing
crops for residues of triflumizole and its aniline
containing metabolites at the proposed tolerance levels. The
analytical method has been independently validated. Residue
levels of triflumizole are converted to FA-1-1 by acidic and
alkaline reflux, followed by distillation. Residues are then
extracted and subjected to solid phase extraction (SPE) purification.
Detection and quantitation are conducted by a GC equipped
with nitrogen phosphorus detector, electron capture detector
or mass spectrometry detection. The LOQ of the method has
been determined at 0.05 ppm for the combined residues of triflumizole
and FA-1-1 in mustard greens. The enforcement methodology
has been submitted to the Food and Drug Administration (FDA)
for publication in the PAM II. |
| March
28, 2007 |
EPA-HQ-OPP-2006-0115 |
Pesticide
Registration Review; New Docket Opened for Review and Comment.
As directed by FIFRA section 3(g), EPA is periodically reviewing
pesticide registrations to assure that they continue to satisfy
the FIFRA standard for registration--that is, they can still
be used without unreasonable adverse effects on human health
or the environment. The implementing regulations establishing
the procedures for registration review appear at 40 CFR part
155. A pesticide's registration review begins when the Agency
establishes a docket for the pesticide's registration review
case and opens the docket for public review and comment. At
present, EPA is opening registration review for triflumizole.
Comments must be received on or before
June 26, 2007.
| DOCUMENTS IN FEDERAL REGISTER
DOCKET |
Triflumizole Summary Document including
Preliminary Work Plan Document (51
pages)
EPA-HQ-OPP-2006-0115-0003 |
List of Products Containing Triflumizole
Pesticide Products Registered Under Section 3 of FIFRA
(1 page)
EPA-HQ-OPP-2006-0115-0004 |
| HEALTH EFFECTS
DOCUMENTS |
Review of Triflumizole Incidents Reports.
August 15, 2006 (3
pages)
EPA-HQ-OPP-2006-0115-0005 |
Section 18 Specific Exemption for the
Use of Triflumizole on Parsley, Dandelion, Swiss Chard,
Collards, Kale, Kohlrabi, Mustard Greens, Chinese Napa
Cabbage, Broccoli, Cilantro, and Turnip Greens in Texas.
March 1, 2006. (12
pages)
EPA-HQ-OPP-2006-0115-0006 |
Triflumizole – Exposure/Risk Assessment
for the FIFRA Section 18 Use of Triflumizole on Leafy
Green Vegetable in Texas. February 1, 2006. (10
pages)
EPA-HQ-OPP-2006-0115-0007 |
Request for Permanent Tolerance for Residues
of Triflumizole in/on Imported Greenhouse-Grown Tomatoes
and Section 18 Specific Exemption for the Use of Triflumizole
on Turnip Greens in Texas. May 26, 2005.
(12 pages)
EPA-HQ-OPP-2006-0115-0008 |
Section 18 Specific Exemptions for the
Use of Triflumizole on parsley, Dandelion, Swiss Chard,
Collards, Kale, Kohlrabi, Mustard Greens, Chinese Napa
Cabbage, Broccoli, and Cilantro. January 28, 2005. (14
pages)
EPA-HQ-OPP-2006-0115-0009 |
Triflumizole – Waiver Request for
28-Day Inhalation Study September 23, 2004. (3
pages)
EPA-HQ-OPP-2006-0115-0010 |
Triflumizole – Amendment to: Human
Exposure and Risk Assessment for Proposed New Uses on
Cherries, Hazelnuts, Cucurbits, Strawberries, Outdoor
Ornamentals, and Institutional, Recreational and Residential
Landscapes. April 26, 2002. (15
pages)
EPA-HQ-OPP-2006-0115-0011 |
Triflumizole in/on Strawberries, Cucurbit
Vegetables, Cherries, and Hazelnuts. April 25, 2002. (44
pages)
EPA-HQ-OPP-2006-0115-0012 |
Minutes of 9/17/03 Chem SAC Meeting.
1/20/04. Render Food/Non-Food use Decision on the Proposed
Use of Procure 50WS Fungicide to Treat Pineapple Seed
Pieces. January 20, 2003. (1
page)
EPA-HQ-OPP-2006-0115-0013 |
HED SOP 2002.01 Standard Operating Procedure
– Waiver Criteria for Multiple-Exposure Inhalation
Toxicity Studies August 15, 2002. (7
pages)
EPA-HQ-OPP-2006-0115-0014 |
Triflumizole – Revised Report of
the Hazard Identifiaction assessment Review committee.
March 19, 2002. (30 pages)
EPA-HQ-OPP-2006-0115-0015 |
Triflumizole – Report of the FQPA
Safety Factor Committee. March 21, 2002. (9
pages)
EPA-HQ-OPP-2006-0115-0016 |
Triflumizole. Issues to be Presented
to the Health Effects Division (HED) Metabolism Assessment
Review Committee (MARC) Meeting on 2/26/2002. February
26, 2002. (24
pages)
EPA-HQ-OPP-2006-0115-0017 |
| ECOLOGICAL
FATE & EFFECTS DOCUMENTS |
Section 3 Request for Uses of Triflumizole
on Foliage for Filberts. February 7, 2006. (130
pages)
EPA-HQ-OPP-2006-0115-0018 |
Tier 1 Drinking Water Assessment for
Use of Triflumizole on Filberts. January 5, 2006.
(8 pages)
EPA-HQ-OPP-2006-0115-0019 |
Tier 1 Estimated Drinking Water Concentrations
for Triflumizole including Degradates. March 13, 2002.
(14 pages)
EPA-HQ-OPP-2006-0115-0020 |
Section 3 Request for Use of Triflumizole
(Chemical #128879) on Cherries, Strawberries, Cucurbits,
and Ornamentals. May 21, 2001. (44
pages)
EPA-HQ-OPP-2006-0115-0021 |
Triflumizole Screening Level Estimated
of Agricultural Uses March 29, 2006. (2
pages)
EPA-HQ-OPP-2006-0115-0022 |
Triflumizole Appendix A. October 26,
2006. (3
pages)
EPA-HQ-OPP-2006-0115-0023 |
|
| August 23, 2006 |
EPA-HQ-OPP-2006-0461 |
Pesticide
tolerance. FINAL
RULE.
This regulation establishes time-limited tolerances for
combined residues of triflumizole in or on turnip, greens. Additionally,
this regulation increases the tolerance levels and extends time-limited
tolerances for Chinese Napa cabbage, collards, coriander leaves
(cilantro), kale, kohlrabi, mustard greens and parsley. Further,
this regulation extends time-limited tolerances for broccoli,
dandelion and Swiss chard.
| Commodity |
PPM |
Expiration
date |
| Broccoli |
1.0 |
12/31/09 |
| Cabbage,
chinese, napa |
20 |
12/31/09 |
| Collards |
20 |
12/31/09 |
| Coriander,
leaves |
20 |
12/31/09 |
| Dandelion,
leaves |
7.0 |
12/31/09 |
| Kale |
20 |
12/31/09 |
| Kohlrabi |
20 |
12/31/09 |
| Mustard,
greens |
20 |
12/31/09 |
| Parsley,
leaves |
20 |
12/31/09 |
| Swiss
chard |
7.0 |
12/31/09 |
| Turnip,
greens |
20 |
12/31/09 |
|
| March
15, 2006 |
EPA-HQ-OPP-2006-0103 |
IR-4.
Pesticide Tolerance.
FINAL RULE. (Pesticide
Petition 3E6535).
This regulation establishes a tolerance for combined residues
of triflumizole and its metabolites
containing the 4-chloro-2-trifluoromethylaniline moiety,
calculated as the parent compound in or on filberts
at 0.05 ppm.
-- A
summary of the toxicological endpoints for triflumizole used
for human risk assessment is discussed in Unit VI.A. of the
final rule published in the Federal Register - SEE
BELOW - April 8, 2005.
-- Specific
information on the studies received and the nature of the
toxic effects caused by triflumizole as well as the no observed
adverse effect level (NOAEL) and the lowest observed adverse
effect level (LOAEL) from the toxicity studies published in
the Federal Register - SEE
BELOW - June 12, 2002.
|
| Jan
18, 2006 |
EPA-HQ-OPP-
2005-0547 |
IR-4.
Amendment
to Existing Tolerance. PP 3E6535.
IR-4, 681 U.S. Highway #1 South, North Brunswick, NJ 08902,
proposes to amend 40 CFR 180.476 by establishing tolerances
for the combined residues of the fungicide triflumizole and
its metabolites containing the 4-chloro-2-trifluoromethylaniline
moiety in or on the food commodity filberts
at 0.05 parts per million (ppm). The analytical method
is suitable for analyzing crops for residues of triflumizole
and its aniline containing metabolites at the proposed tolerance
levels. The analytical method has been independently validated.
Residue levels of triflumizole are converted to FA-1-1 by acidic
and alkaline reflux, followed by distillation. Residues are
then extracted and subjected to SPE purification. Detection
and quantitation are conducted by a gas chromatography equipped
with nitrogen phosphorus detector, electron capture detector
or mass spectrometry detector. The limit of quantitation of
the method has been determined at 0.05 ppm for the combined
residues of triflumizole and FA-1-1 in filberts. The enforcement
methodology has been submitted to the Food and Drug Administration
for publication in the Pesticide Analytical Manual, Vol. II
(PAM II). |
| April
8, 2005 |
OPP-2005-0054 |
Pesticide
Tolerances for Emergency Exemptions in TEXAS. FINAL
RULE.
EPA, on its own initiative, in accordance with sections 408(e)
and 408 (l)(6) of the Federal Food, Drug, and Cosmetic Act
(FFDCA), 21 U.S.C. 346a, is establishing time-limited tolerances
for combined residues of the fungicide triflumizole and
its metabolites containing the 4-chloro-2-trifluoromethylaniline
moiety, calculated as the parent compound, in or on
the following commodities.Texas
has declared a crisis exemption under FIFRA section 18 for
the use of triflumizole in or on:
| Commodity |
Parts
per
million |
Expiration/
revocation date |
| Broccoli |
1.0 |
6/30/08 |
| Cabbage,
chinese, napa |
9.0 |
6/30/08 |
| Collards |
9.0 |
6/30/08 |
| Coriander,
leaves (cilantro) |
9.0 |
6/30/08 |
| Dandelion,
leaves |
7.0 |
6/30/08 |
| Kale |
9.0 |
6/30/08 |
| Kohlrabi |
9.0 |
6/30/08 |
| Mustard
greens |
9.0 |
6/30/08 |
| Parsley,
leaves |
9.0 |
6/30/08 |
| Swiss
chard |
7.0 |
6/30/08 |
Although
these tolerances will expire and are revoked on June 30, 2008,
under section 408(l)(5) of the FFDCA, residues of the pesticide
not in excess of the amounts specified in the tolerances remaining
in or on these commodities after that date will not be unlawful,
provided the pesticide is applied in
a manner that was lawful under FIFRA, and the residues do
not exceed a level that was authorized by these tolerances
at the time of that application.
In
the Agency's previous triflumizole human health risk assessment,
the following toxicity studies were determined to be data
gaps:
• A 28-day rat inhalation study Guideline Number (GLN)
870.3465)),
• acute rat neurotoxicity study (GLN 870.6200), and
• subchronic rat neurotoxicity study (GLN 870.6200).
As acceptable
acute and sub-chronic neurotoxicity studies have been submitted,
the Agency has determined that the 3x
UFDB should be removed from the acute and chronic RfDs. In
addition, the FQPA SFC recommended a special FQPA SF
[safety factor] be reduced to 1x. The
Agency has re-evaluated the quality of the exposure and hazard
data; and, based on these data, concluded that the special
FQPA SF remain at 1x.
Conditions
The petitioner should submit adequate
limited field rotational crop data on wheat at plant-back
intervals longer than 120 days. Alternatively, the petitioner
has the option of submitting a full set of residue field trials
on all intended rotational crops other than leafy and root
vegetables.
Excerpts
from Table 1: Summary
of Toxicological Dose and Endpoints for Triflumizole for Use
in Human Risk Assessment:
Acute
Dietary (females 13-50 years of age).
Developmental Toxicity Study - Rat
Developmental LOAEL = 35 mg/kg/day based on decreased
numbers of viable fetuses,
increased dead or resorbed fetuses,
increased numbers of late resorptions,
decreased fetal body weight,
and increased incidences of cervical
ribs.
Acute
Dietary (general U.S. population) (including infant and children).
Acute Neurotoxicity Study - Rat
LOAEL = 100 mg/kg/day based on functional observational battery
findings (neuromuscular impairment)
and decreased locomotor activity.
Chronic
Dietary (all populations).
Multi-generation Reproduction Study - Rat
Reproductive LOAEL = 3.5 mg/kg/day based on increased
gestation length in dams of the F3a
interval.
Short-Term
Oral (1-30 days) (Residential).
Multi-generation Reproduction Study - Rat
Reproductive LOAEL = 3.5 mg/kg/day based
on increased gestation length in
dams of the F3a interval.
Intermediate-Term
Oral (1-6 months) (Residential).
Multi-generation Reproduction Study - Rat
LOAEL = 21 mg/kg/day, based on decreased
body weight gain in pups during lactation
and decreased body weight and
body weight gain in parental animals.
Short-Term
Dermal (1-30 days) (Occupational/Residential).
Multi-generation Reproduction Study -
Rat
LOAEL = 21 mg/kg/day, based on decreased
body weight gain in pups during lactation.
Intermediate-
and Long-Term Dermal (1- 6 months and 6 month or longer) (Occupational/Residential).
Multi-generation
Reproduction Study - Rat
LOAEL = 3.5 mg/kg/day based on
increased gestation length in
the dams of the F3a interval.
Short-Term
Inhalation (1-30 days) (Occupational/Residential).
Multi-generation
Reproduction Study - Rat
LOAEL = 21 mg/kg/day, based on decreased
body weight gain in pups during lactation.
Intermediate-
and Long-Term Inhalation (1-6 months and 6 month or longer)
(Occupational/Residential).
Multi-generation Reproduction Study - Rat
LOAEL = 3.5 mg/kg/day based on
increased gestation length in
the dams of the F3a interval. |
| Feb
23, 2005 |
OPP-2005-0037 |
Pesticide
Emergency Exemptions.
Texas Department of Agriculture
Crisis: On December 21, 2004, for the use of triflumizole on
parsley, dandelion, Swiss chard, collards,
kale, kohlrabi, mustard greens, napa cabbage, and cilantro
to control powdery mildew. This program is expected to end on
October 1, 2005. |
| Feb
10, 2005 |
OPP-2005-0025 |
Removal
of Expired Time-limited Tolerances for Emergency Exemptions.
FINAL
RULE.
The time-limited tolerance for filbert
is being removed from Sec. 180.476 because it expired
on June 30, 2004. |
| Aug
23, 2002 |
OPP-2002-0183 |
Pesticide
Tolerance for Emergency Exemption; in or on Filbert at 0.05
ppm - for
the state of Oregon only. FINAL RULE. Refer
to the June 12,
2002 Federal Register document for a detailed discussion
of the aggregate risk assessments and determination of safety.
EPA relies upon that risk assessment and the findings made
in the Federal Register document in support of this action
- see excerpts below. This tolerance will expire and is revoked
on June 30, 2004. |
| June
12, 2002 |
OPP-2002-0063 |
UNIROYAL
-Pesticide Tolerance. FINAL
RULE. This
regulation establishes tolerances for the combined residues
of triflumizole and its metabolites containing the 4-chloro-2-trifluoromethylaniline
moiety, calculated as the parent compound in or on cucurbit
Group 9 vegetables (0.5 ppm), strawberries (2 ppm), sweet
cherries (1.5 ppm) , and tart cherries (1.5 ppm).
-- 90-Day oral NOAEL = Males: toxicity
rodents 15.3 mg/kg/day; (rat) Females: 17.2 mg/ kg/day LOAEL
= Males: 176.5 mg/kg/day; Females: 217.9 mg/ kg/day based
on increased kidney and liver weights and
the accumulation of fat droplets in the liver.
-- Prenatal Maternal developmental in
NOAEL = 10 mg/kg/ rodents (rat) day LOAEL = 35 mg/kg/ day
based on decreased body weight gain and food consumption,
and increased placental, spleen and liverweights. Developmental
NOAEL = 10 mg/kg/ day LOAEL = 35 mg/kg/ day based on decreased
numbers of viable fetuses, increased
dead or resorbed fetuses, increased
numbers of late resorptions, decreased
fetal body weight, and increased incidences of cervical
ribs.
-- Prenatal Maternal developmental in
NOAEL = 50 mg/kg/ nonrodents day (rabbit) LOAEL = 100 mg/kg/
day based on decreased body weight gains, food consumption,
and placental weights. Developmental
NOAEL = 50 mg/kg/ day LOAEL = 100 mg/kg/ day based on
decreased 24-hour survival, decreased
placental weights, and increased
fetal and litter incidences of
lumbar ribs.
-- Reproduction and Parental/Systemic
fertility effects NOAEL = 8.5 mg/kg/ (rat) day LOAEL = 21
mg/kg/ day based on decreased body weight and overall body
weight gain, increased relative liver weights,
and increased mortality in females. Reproductive
NOAEL = not identified LOAEL = 3.5 mg/kg/ day based on
increased gestation length in P. Offspring
NOAEL = 8.5 mg/kg/ day LOAEL = 21 mg/kg/ day based on decreased
pup body weight,survival indices, and litter sizes and a slight
increased incidence of hydronephrosis
in F1a pups.
-- Reproduction and Parental/Systemic
fertility effects NOAEL = 8.5 mg/kg/ (rat) day LOAEL = not
established Reproductive NOAEL = 1.5 mg/kg/ day LOAEL = 3.5
mg/kg/ day based on based on increased gestation length
in dams of the F3a interval. Offspring NOAEL =
3.5 mg/kg/ day LOAEL = 8.5 mg/kg/ day based on decreased pup
weights,survival indices, and litter sizes in both F3
litters, reduced litter size in the F1a litter,
increased total-litter mortality
in the F3a litter, and developmental effects
in theF1b and F2b progeny.
-- Chronic toxicity NOAEL = Males: nonrodents
(dog) 10.00 mg/kg/day; Females: 10.69 mg/ kg/day LOAEL = Males:
34.10 mg/kg/day; Females: 35.17 mg/ kg/day based on increased
alkaline phosphatase activity and a
mild, macrocytic anemia in males, increased
absolute and relative liver weights
in both sexes, and on macroscopic findings in the liver
of both sexes.
-- Carcinogenicity NOAEL = Males: (mouse)
16.2 mg/kg/day; Females: 21.7 mg/ kg/day LOAEL = Males: 67.4
mg/kg/day; Females: 86.1 mg/ kg/day based on microscopic lesions
of the liver. No evidence of
carcinogenicity |
| Nov
14, 2001 |
OPP-181082 |
- Denial
of requests for Pesticide Emergency Exemptions. EPA
denied the following:
- Idaho:
On June 26, 2001, EPA denied the use of triflumizole on
sweet cherries to control powdery mildew. This request was
denied because the claim of resistance to registered alternatives
was not fully substantiated.
- Oregon:
On February 1, 2001, EPA denied the use of triflumizole
on hazelnut to control Eastern Filbert Blight (EFB). This
request was denied because the submission did not meet the
criteria for an urgent, non-routine situation based on the
availability of an effective alternative, tebuconazole,
available under a FIFRA section 18 exemption.
- Oregon:
On February 1, 2001, EPA denied the use of triflumizole
on hazelnut to control Eastern Filbert Blight (EFB). This
request was denied because the submission did not meet the
criteria for an urgent, non-routine situation based on the
availability of an effective alternative, tebuconazole,
available under a FIFRA section 18 exemption.
- Utah:
On June 26, 2001, EPA denied the use of triflumizole on
cherries to control powdery mildew. This request was denied
because the claim of resistance to registered alternatives
was not fully substantiated.
- Washington:
On June 26, 2001, EPA denied the use of triflumizole on
sweet cherries to control powdery mildew. This request was
denied because the claim of resistance to registered alternatives
was not fully substantiated.
|
|
July 6, 2001 |
PF-1031 |
UNIROYAL
- Petition
to establish a tolerance for residues in or on strawberries
at 2.0 ppm, the cucurbit crop group at 0.5 ppm, and cherries
at 2.0 ppm. |
| Oct
3, 1997 - FINAL |
NA |
Cut
Roses. Exception Decisions to Early Entry Prohibition, Worker
Protection Standard; Technical Amendment. - FINAL
RULE. See Federal Register of Oct 30, 1996 below.
|
| Oct
3, 1997- Proposed |
OPP-250121 |
Cut
Roses. Pesticide Worker Protection Standard; Administrative
Exception for Cut-Rose Hand Harvesting; Administrative Decision.
Proposed Rule. |
| Augt
4, 1997 |
OPP-300523 |
Pesticides
Subject to Tolerance Reassessment. |
| July
16, 1997 |
NA |
UNIROYAL
- Request
to voluntarily cancel certain pesticide registration for:
Terraguard 50W. EPA Reg. No. 000400 OR-91-000 |
| Oct
30, 1996 |
OPP-300164I |
Cut-Roses;
Request for Exception to Worker Protection Standard's Prohibition
of Early Entry into Pesticide-Treated Areas to Harvest Roses
by Hand Cutting. |
| Nov
16, 1994 |
PP
6F3372
and
FAP 6H5497/R2085 |
UNIROYAL
- Pesticide
Tolerances and Feed Additive Regulations.- FINAL
RULE. Tolerances are established for the combined
residues of the fungicide triflumizole and its metabolites
containing the 4-chloro-2-trifluoromethylaniline moiety in
or on the following raw agricultural commodities: Apples and
Pears at 0.5 ppm; Grapes at 2.5 ppm; Meat and fat of horses,
hogs, goats, sheep, and poultry at 0.05 ppm.
Tolerances
are established for the combined residues of the fungicide
triflumizole and its metabolites containing the 4-chloro-2-trifluoromethylaniline
moiety, calculated as the parent compound, in or on the following
processed feed commodities when present therein as a result
of application to growing crops: Grape pomace at 15 ppm; Raisin
waste at 10 ppm; Apple pomace at 2 ppm. |
| 1993 |
NA |
UNIROYAL
- Petition to
Establish Tolerances for the combined residues of the fungicide
in or on the following commodities: apples at 0.1 ppm;
cattle, fat, meat and meat byproducts (mbyp) at 0.05 ppm;
grapes at 0.3 ppm; hogs, fat, meat and mbyp at 0.05 ppm; milk
at 0.05 ppm; pears at 0.1 ppm; and poultry, eggs, fat, meat
and mbyp at 0.05 ppm. Note. The Fed. Reg. does not have online
access to its 1993 edition. This notice comes from the Pesticide
Managment Education Program at Cornell U. |
|