|
Return
to Flufenpyr-ethyl
Index Page
Activity:
Herbicide
(Pyridazinone)
Structure
for "Flufenpyr":
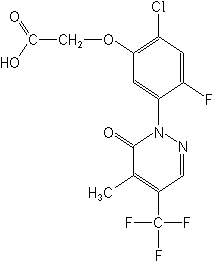
Adverse Effects:
Anemia
Blood
Endocrine: Thymus
Kidney
Liver
September
19, 2003:
US EPA approved the first-time tolerances
for residues of Flufenpyr-ethly in or on food commodities.
No environmental studies are available.
The new tolerances are:
Corn,
field, grain - 0.01 ppm
Corn, field, forage - 0.05 ppm
Corn, field, stover - - 0.05 ppm
Soybean, seed - 0.01 ppm
Sugarcane, cane - 0.01 ppm
Ref: Federal Register, Flufenpyr-Ethyl;
Pesticide Tolerance. Final Rule. September 19, 2003.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.sept2003.htm
|
Anemia
(click on for all fluorinated pesticides)
-- 870.4200. Carcinogenicity
rodents (mouse). NOAEL = 39.9 - 43.7 mg/kg/day M/F LOAEL = 401.8
- 447.9 mg/kg/day M/F, based on liver toxicity in both sexes and
mild anemia in males No evidence
of carcinogenicity.
Ref:
Federal Register: September 19, 2003. Flufenpyr-Ethyl; Pesticide
Tolerance. Final Rule.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.sept2003.htm
--
Mice. In a 4-week study, CD-1 mice
were fed pure flufenpyr- ethyl at dose levels of 0, 300, 1,000,
3,000, and 7,000 ppm. Effects were slight
anemia, changes in blood biochemistry...
-- Mice.
In a 13-week study, flufenpyr-ethyl technical was administered
to mice at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm.
Slight anemia and
blood biochemistry changes
were noted...
Ref:
Federal Register: June 25, 2003 (Volume 68, Number 122)] [Notices]
[Page 37813-37820]. Flufenpyr-ethyl; Notice of Filing a Pesticide
Petition to Establish a Tolerance for a Certain Pesticide Chemical
in or on Food.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.june.03.htm
Blood
(click
on for all fluorinated pesticides)
-- EPA has not had
the opportunity to review the toxicity studies on flufenpyr-ethyl
and has not established toxic endpoints.
-- Subchronic toxicity. Subchronic oral toxicity studies conducted
with flufenpyr-ethyl in the rat, mouse and dog indicate a low
level of toxicity. i. Rats. Pure (99.4%) flufenpyr-ethyl was tested
in rats at dose levels of 0, 600, 2,000, 6,000, and 20,000 ppm
in the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, slight
hematological and blood biochemistry changes...
-- In an additional study, flufenpyr-ethyl technical was tested
in rats at dose levels of 0, 1,000, 10,000, and 20,000 ppm in
the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, and mild urinalysis, hematological
and blood biochemistry changes...
-- Mice. In a 4-week study, CD-1 mice were fed pure flufenpyr-
ethyl at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm. Effects
were slight anemia, changes in blood biochemistry...
-- In a 13-week study, flufenpyr-ethyl technical was administered
to mice at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm.
Slight anemia and blood biochemistry changes
were noted...
-- Mice. In a 78-week oncogenicity study with mice, flufenpyr-
ethyl technical was administered at dose levels of 0, 350, 3,500,
and 7,000 ppm. Male animals exhibited slight
anemia. Females had increased liver
and kidney weights (week 53 only)...
Ref:
Federal Register: June 25, 2003 (Volume 68, Number 122)] [Notices]
[Page 37813-37820]. Flufenpyr-ethyl; Notice of Filing a Pesticide
Petition to Establish a Tolerance for a Certain Pesticide Chemical
in or on Food.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.june.03.htm
Endocrine:
Thymus (click
on for all fluorinated pesticides)
-- EPA has not had
the opportunity to review the toxicity studies on flufenpyr-ethyl
and has not established toxic endpoints.
-- In an additional study, flufenpyr-ethyl technical was tested
in rats at dose levels of 0, 1,000, 10,000, and 20,000 ppm in
the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, and mild urinalysis, hematological
and blood biochemistry changes. Thymus weights
were slightly increased...
-- Mice. In a 4-week study, CD-1 mice were fed pure flufenpyr-
ethyl at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm. Effects
were slight anemia, changes in blood biochemistry, increased liver
and thymus weights, and enlarged
liver...
Ref:
Federal Register: June 25, 2003 (Volume 68, Number 122)] [Notices]
[Page 37813-37820]. Flufenpyr-ethyl; Notice of Filing a Pesticide
Petition to Establish a Tolerance for a Certain Pesticide Chemical
in or on Food.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.june.03.htm
Kidney
(click
on for all fluorinated pesticides)
-- The kidney
and liver appear to be the target organs of flufenpyr-ethyl.
EPA has not had the opportunity to review the toxicity studies
on flufenpyr-ethyl and has not established toxic endpoints.
-- Reproduction. In the rat reproduction study, flufenpyr-ethyl
technical was administered in the diet at levels of 0, 200, 2,000,
and 20,000 ppm for 2-generations. Parental toxicity was observed
at all dose levels, although the effects at the low dose were
minimal. Parental toxicity was exhibited by dose-related microscopic
changes in the kidney in high dose F0 animals, in all treated
F1 males, and in high dose F1 females. There were also 2 high
dose F1 males that died possibly as a result of treatment...
-- A second 1-generation reproduction study was performed to establish
a clear NOAEL for adult
kidney lesions using the dose levels of 20, 50 and 100
ppm. The results of the study indicate that the NOAEL for histological
changes in the kidneys of F1 male rats was 100 ppm. No
other treatment related findings were noted at any dose level
indicating 100 ppm as the NOAEL for treatment and reproductive
effects evaluated in the study.
-- A mechanistic study was also conducted to investigate the reproducibility
and reversibility of the kidney lesions
observed in the initial 2-generation reproduction study. In the
first study, the effects observed at 200 ppm in the F1 males,
basophilic tubules and interstitial inflamation, were minimal
but slightly increased in incidence and severity and a slight
increase in interstitial fibrosis of the cortex was also observed.
In this mechanistic study, using dose levels of 0 and 2,000 ppm,
the NOAEL for histological changes in the kidneys of F0 and F1
male rats and reproductive effects was 2,000 ppm. The histological
changes seen in the kidneys in the original study was not reproducible.
-- Subchronic toxicity. Subchronic oral toxicity studies conducted
with flufenpyr-ethyl in the rat, mouse and dog indicate a low
level of toxicity. i. Rats. Pure (99.4%) flufenpyr-ethyl was tested
in rats at dose levels of 0, 600, 2,000, 6,000, and 20,000 ppm
in the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, slight hematological and
blood biochemistry changes, decreased spleen weights, an increase
in the incidence and severity of basophilic tubules of the kidneys
and slight to mild diffusely distributed vacuolation in the
liver. Based on these results, the NOAEL was 2,000 ppm
(134.2 mg/kg/day) for the males and 20,000 ppm (1,509.6 mg/kg/day)
for the females.
-- Mice. In a 78-week oncogenicity study with mice, flufenpyr-
ethyl technical was administered at dose levels of 0, 350, 3,500,
and 7,000 ppm. Male animals exhibited slight anemia. Females had
increased liver and kidney weights
(week 53 only). Slight to moderate hepatocellular fatty vacuolation
and necrosis were observed. There were no increases in incidence
of pre-neoplastic or neoplastic lesions. Based on these results,
the NOAEL was 350 ppm for both sexes (39.9 mg/ kg/day for males
and 43.7 mg/kg/day for females).
Ref:
Federal Register: June 25, 2003 (Volume 68, Number 122)] [Notices]
[Page 37813-37820]. Flufenpyr-ethyl; Notice of Filing a Pesticide
Petition to Establish a Tolerance for a Certain Pesticide Chemical
in or on Food.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.june.03.htm
Liver
(click on for all fluorinated pesticides)
Carcinogenicity
rodents (mouse) - [870.4200]. NOAEL = 39.9 - 43.7 mg/kg/day M/F
LOAEL = 401.8 - 447.9 mg/kg/day M/F, based on liver
toxicity in both sexes and mild anemia in males. No evidence
of carcinogenicity.
Ref: Federal Register: September 19, 2003.
Flufenpyr-Ethyl; Pesticide Tolerance. Final Rule.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.sept2003.htm
-- The kidney
and liver appear to be the target organs of flufenpyr-ethyl.
EPA has not had the opportunity to review
the toxicity studies on flufenpyr-ethyl and has not established
toxic endpoints.
-- Reproduction. In the rat reproduction study, flufenpyr-ethyl
technical was administered in the diet at levels of 0, 200, 2,000,
and 20,000 ppm for 2-generations. Parental toxicity was observed
at all dose levels, although the effects at the low dose were
minimal. Parental toxicity was exhibited by dose-related
microscopic changes in the kidney in high dose F0 animals,
in all treated F1 males, and in high dose F1 females. There were
also 2 high dose F1 males that died possibly as a result of treatment.
Midzonal cytoplasmic vacuolation of the
hepatocytes was also observed in the liver of all groups of treated
animals in both generations. Based on the results of this
study, the NOAEL for parental toxicity was considered to be less
than 200 ppm. The NOAEL for reproductive and neonatal toxicity
was considered to be 20,000 ppm.
-- Subchronic toxicity. Subchronic oral toxicity studies conducted
with flufenpyr-ethyl in the rat, mouse and dog indicate a low
level of toxicity. i. Rats. Pure (99.4%) flufenpyr-ethyl was tested
in rats at dose levels of 0, 600, 2,000, 6,000, and 20,000 ppm
in the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, slight hematological and
blood biochemistry changes, decreased spleen weights, an
increase in the incidence and severity of basophilic tubules of
the kidneys and slight to mild diffusely distributed vacuolation
in the liver. Based on these results,
the NOAEL was 2,000 ppm (134.2 mg/kg/day) for the males and 20,000
ppm (1,509.6 mg/kg/day) for the females.
-- In an additional study, flufenpyr-ethyl technical was tested
in rats at dose levels of 0, 1,000, 10,000, and 20,000 ppm in
the diet for 13 weeks. Effects observed included urinary incontinence,
increased food and water consumption, and mild urinalysis, hematological
and blood biochemistry changes. Thymus weights were slightly increased.
Diffusely distributed hepatic vacuolation
was seen in the high dose males. Based on these findings, the
NOAEL was 10,000 ppm (595.2 mg/kg/ day) in the males and 20,000
ppm (1,377.5 mg/kg/day) in the females.
-- Mice. In a 4-week study, CD-1 mice were fed pure flufenpyr-
ethyl at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm. Effects
were slight anemia, changes in blood biochemistry, increased liver
and thymus weights, and enlarged
liver. Centrilobular hepatocellular
hypertrophy and vacuolation and increases in the severity
and incidence of hepatic focal and single
cell necrosis were observed. Based on these findings, the
NOAEL was 300 ppm (44.9 mg/kg/day) for males
and 1,000 (210.5 mg/kg/day) for females.
-- In a 13-week study, flufenpyr-ethyl technical was administered
to mice at dose levels of 0, 300, 1,000, 3,000, and 7,000 ppm.
Slight anemia and blood biochemistry changes were noted. Liver
weights were increased and ovary weights were decreased.
Histopathological findings included: Hepatocellular
fatty vacuolation. The NOAEL for this study in both sexes
was [[Page 37818]] 1,000 ppm (128.4 mg/kg/day for males and 155.7
mg/kg/day for females).
-- Mice. In a 78-week oncogenicity study with mice, flufenpyr-
ethyl technical was administered at dose levels of 0, 350, 3,500,
and 7,000 ppm. Male animals exhibited slight anemia. Females had
increased liver and kidney weights
(week 53 only). Slight to moderate hepatocellular
fatty vacuolation and necrosis were observed. There were
no increases in incidence of pre-neoplastic or neoplastic lesions.
Based on these results, the NOAEL was 350 ppm for both sexes (39.9
mg/ kg/day for males and 43.7 mg/kg/day for females).
Ref:
Federal Register: June 25, 2003 (Volume 68, Number 122)] [Notices]
[Page 37813-37820]. Flufenpyr-ethyl; Notice of Filing a Pesticide
Petition to Establish a Tolerance for a Certain Pesticide Chemical
in or on Food.
http://www.fluorideaction.org/pesticides/flufenpyr-ethyl.fr.june.03.htm
|

