Chapter 7: Neurotoxicity and Neurobehavioral Effects
This chapter evaluates the effects of fluoride on the nervous system and behavior, with particular emphasis on studies conducted since the earlier NRC (1993) review. The human data include epidemiologic studies of populations exposed to different concentrations of fluoride and individual case studies. In addition, laboratory studies of behavioral, biochemical, and neuroanatomical changes induced by fluoride have been reviewed and summarized. At the end of the chapter, conclusions and recommendations for future research are presented.
HUMAN STUDIES
Cognitive Effects
Several studies from China have reported the effects of fluoride in drinking water on cognitive capacities (X. Li et al. 1995; Zhao et al. 1996; Lu et al. 2000; Xiang et al. 2003a,b). Among the studies, the one by Xiang et al. (2003a) had the strongest design. This study compared the intelligence of 512 children (ages 8-13) living in two villages with different fluoride concentrations in the water. The IQ test was administered in a double-blind manner. The high-fluoride area (Wamiao) had a mean water concentration of 2.47 ± 0.79 mg/L (range 0.57-4.50 milligrams per liter [mg/L]), and the low-fluoride area (Xinhuai) had a mean water concentration of 0.36 ± 0.15 mg/L (range 0.18-0.76 mg/L). The populations studied had comparable iodine and creatinine concentrations, family incomes, family educational levels, and other factors. The populations were not exposed to other significant sources of fluoride, such as smoke from coal fires, industrial pollution, or consumption of brick tea. Thus, the difference in fluoride exposure was attributed to the amount in the drinking water. Mean urinary fluoride concentrations were found to be 3.47 ± 1.95 mg/L in Wamiao and 1.11 ± 0.39 mg/L in Xinhuai. Using the combined Raven’s Test for Rural China, the average intelligence quotient (IQ) of the children in Wamiao was found to be significantly lower (92.2 ± 13.00; range, 54-126) than that in Xinhuai (100.41 ± 13.21; range, 60-128).
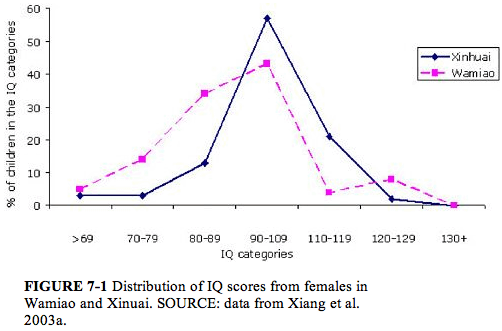
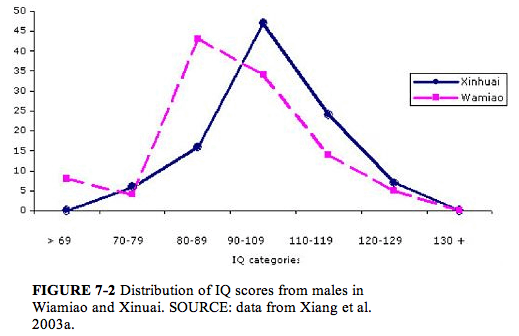
The IQ scores in both males and females declined with increasing fluoride exposure. The distribution of IQ scores from the females in the two villages is shown in Figure 7-1. A comparable illustration of the IQ scores of males is shown in Figure 7-2. The number of children in Wamiao with scores in the higher IQ ranges was less than that in Xinhuai. There were corresponding increases in the number of children in the lower IQ range. Modal scores of the IQ distributions in the two villages were approximately the same. A follow-up study to determine whether the lower IQ scores of the children in Wamiao might be related to differences in lead exposure disclosed no significant difference in blood lead concentrations in the two groups of children (Xiang et al. 2003b).
A study conducted by Lu et al. (2000) in a different area of China also compared the IQs of 118 children (ages 10-12) living in two areas with different fluoride concentrations in the water (3.15 ± 0.61 mg/L in one area and 0.37 ± 0.04 mg/L in the other). The children were lifelong residents of the villages and had similar social and educational levels. Urinary fluoride concentrations were measured at 4.99 ± 2.57 mg/L in the high-fluoride area and 1.43 ± 0.64 mg/L in the low-fluoride area. IQ measurements using the Chinese Combined Raven’s Test, Copyright 2 (see Wang and Qian 1989), showed significantly lower mean IQ scores among children in the high-fluoride area (92.27 ± 20.45) than in children in the low-fluoride area (103.05 ± 13.86). Of special importance, 21.6% of the children in the high-fluoride village scored 70 or below on the IQ scale. For the children in the low-fluoride village, only 3.4% had such low scores. Urinary fluoride concentrations were inversely correlated with mental performance in the IQ test. Qin and Cui (1990) observed similar negative correlation between IQ and fluoride intake through drinking water.
Zhao et al. (1996) also compared the IQs of 160 children (ages 7-14) living in a high-fluoride area (average concentration of 4.12 mg/L) with those of children living in a low-fluoride area (average concentration 0.91 mg/L). Using the Rui Wen Test, the investigators found that the average IQ of children in the high-fluoride area (97.69) was significantly lower than that of children in the low-fluoride area (105.21). No sex differences were found, but, not surprisingly, IQ scores were found to be related to parents’ education. The investigators also reported that enamel fluorosis was present in 86% of the children in the high-exposure group and in 14% of the children in the low-exposure group and that skeletal fluorosis was found only in the high-exposure group at 9%.
Another Chinese study evaluated fluoride exposure due to inhalation of soot and smoke from domestic coal fires used for cooking, heating, and drying grain (Li et al. 1995). Many of the children exhibited moderate to severe enamel fluorosis. The average IQ of 900 children (ages 8-13) from an area with severe enamel fluorosis was 9-15 points lower than the average IQ of children from an area with low or no enamel fluorosis. Urinary fluoride concentrations were found to be inversely correlated with IQ, as measured by the China Rui Wen Scale for Rural Areas, and were monotonically related to the degree of enamel fluorosis. Studies based on fluoride exposure from the inhalation of smoke from coal fires are difficult to interpret because of exposure to many other contaminants in smoke.
The significance of these Chinese studies is uncertain. Most of the papers were brief reports and omitted important procedural details. For example, some studies used a modification of the Raven Progressive Matrix test but did not specify what the modifications were or describe how the test was administered. Most of the studies did not indicate whether the IQ tests were administered in a blinded manner. Some of the effects noted in the studies could have been due to stress induced by the testing conditions. Without detailed information about the testing conditions and the tests themselves, the committee was unable to assess the strength of the studies. Despite this, the consistency of the collective results warrants additional research on the effects of fluoride on intelligence in populations that share similar languages, backgrounds, socioeconomic levels, and other commonalities.
It should be noted that many factors outside of native intelligence influence performance on IQ tests. One factor that might be of relevance to fluoride is impairment of thyroid gland function (see Chapter 8). For example, hypothyroidism produces tiredness, depression, difficulties in concentration, memory impairments, and impaired hearing. In addition, there is some evidence that impaired thyroid function in pregnant women can lead to children with lower IQ scores (Klein et al. 2001).
Mental and Physiological Changes
There are numerous reports of mental and physiological changes after exposure to fluoride from various routes (air, food, and water) and for various time periods (Waldbott et al. 1978). A number of the reports are, in fact, experimental studies of one or more individuals who underwent withdrawal from their source of fluoride exposure and subsequent re-exposures under “blind” conditions. In most cases, the symptoms disappeared with the elimination of exposure to fluoride and returned when exposure was reinstated. In some instances, when the fluoride was given in water, this procedure was repeated several times under conditions in which neither the patient nor the provider of the fluoride knew whether the water contained fluoride. Also reported are instances when fluoride-produced symptoms occurred when people moved into a community with fluoridated water but disappeared when the individuals moved to a nonfluoridated community.
Spittle (1994) reviewed surveys and case reports of individuals exposed occupationally or therapeutically to fluoride and concluded there was suggestive evidence that fluoride could be associated with cerebral impairment. A synopsis of 12 case reports of fluoride-exposed people of all ages showed common sequelae of lethargy, weakness, and impaired ability to concentrate regardless of the route of exposure. In half the cases, memory problems were also reported. Spittle (1994) described several of the biochemical changes in enzymatic systems that could account for some of the psychological changes found in patients. He suggested that behavioral alterations found after excessive exposure could be due to the disruption of the N-H bonds in amines, and subsequently in proteins, by the production of N-F bonds (Emsley et al. 1981). This unnatural bond would distort the structure of a number of proteins with the collective potential to cause important biological effects. Fluorides also distort the structure of cytochrome-c peroxidase (Edwards et al. 1984). Spittle also noted the likelihood of fluoride interfering with the basic cellular energy sources used by the brain through the formation of aluminum fluorides (Jope 1988) and subsequent effects on G proteins.
Effects of Silicofluorides
It has been suggested that the silicofluorides used to fluoridate drinking water behave differently in water than other fluoride salts (see Chapter 2 for further discussion) and produce different biological effects. For example, adding sodium silicofluoride (Na2SiF6) or fluorosilicic acid (H2SiF6) to drinking water has been reported to increase the accumulation of the neurotoxicant lead in the body (Masters and Coplan 1999; Masters et al. 2000). This association was first attributed to increased uptake of lead (from whatever source) caused by fluoride. However, enhanced lead concentrations were found only when the water treatments were made with a fluorosilicate and in children already in a high-lead exposure group.
Urbansky and Schock (undated, 2000) took exception to almost all aspects of the studies by Masters and Coplan on the fluorosilicates. They argued that, under the conditions prevailing at the time of the addition of silicofluorides to drinking water, silicofluorides would be completely hydrolyzed before they reached the consumer’s tap (Urbansky and Schock 2000). Measurement techniques and statistical methods were also questioned. They concluded that there is no “credible evidence” that water fluoridation has any quantifiable effect on the solubility, bioavailability, or bioaccumulation of any form of lead.
Another issue that has been raised about differential effects of silicofluorides comes from the dissertation of Westendorf (1975). In that study, silicofluorides were found to have greater power to inhibit the synthesis of cholinesterases, including acetylcholinesterase, than sodium fluoride (NaF). For example, under physiological conditions, one molar equivalent of silicofluoride is more potent in inhibiting acetylcholinesterase than six molar equivalents of NaF (Knappwost and Westendorf 1974). This could produce a situation in which acetylcholine (ACh) accumulates in the vicinity of ACh terminals and leads to excessive activation of cholinergic receptors in the central and peripheral nervous system. At high concentrations, agents with this capability are frequently used in insecticides and nerve gases. At intermediate concentrations, choking sensations and blurred vision are often encountered. Modifications of the effectiveness of the acetylcholinergic systems of the nervous system could account for the fact that, even though native intelligence per se may not be altered by chronic ingestion of water with fluoride ranging from 1.2 to 3 mg/L, reaction times and visuospatial abilities can be impaired. These changes would act to reduce the tested IQ scores. Such noncognitive impairments in children were reported in a meeting abstract (Calderon et al. 2000), but a full publication has not been issued. Extended reaction times have been associated with impaired function of the prefrontal lobes, a behavioral change not directly tied to alterations in IQ (Winterer and Goldman 2003). Because almost all IQ tests are “time-restricted,” slow reaction times would impair measured performance.
An interesting set of calculations was made by Urbansky and Schock (undated)—namely, compilation of the binding strengths of various elements with fluorine. They studied eight different complexes. Aluminum and fluorine have the highest binding affinity. Fluorine also forms complexes with other elements including sodium, iron, calcium, magnesium, copper, and hydrogen. Associations with some of these other elements may have implications for some of the neurotoxic effects noted after fluoride or SiF exposure.
Dementia
For more than 30 years it has been known that Alzheimer’s disease is associated with a substantial decline in cerebral metabolism (Sokoloff 1966). This original observation has been replicated many times since then. The decrease is reflected in the brain’s metabolic rate for glucose, cerebral rate for oxygen, and cerebral blood flow. In terms of reduced cerebral blood flow, the reduction found in Alzheimer’s patients is about three times greater than in patients with multi-infarct dementia. As early as 1983, Foster et al. (1983) demonstrated a general decline in the rate of utilization of glucose with the marker F-2-fluorodeoxyglucose with a positron-emission tomography scan. Recently, over and above the general decline in aerobic metabolism, several patterns of enhanced decreases in energy utilization have been demonstrated. The temporal, parietal, and frontal regions are areas with some of the greatest reductions (Weiner et al. 1993; Starkstein et al. 1995). It is possible that the decline in glucose utilization is an early sign of the onset of dementia (Johnson et al. 1988; Silverman and Small 2002). In addition there is evidence from a number of sources that alterations induced by Alzheimer’s disease can be observed in many body regions and in blood. This indicates that the disease has system-wide effects in the body. One system particularly sensitive to carbohydrate utilization is the collection of areas involved with the synthesis of ACh. The release of this transmitter is also negatively affected by the interruption of aerobic metabolism and the effect can be noticed in the projection fields of the cholinergic systems. Fluoride produces additional effects on the ACh systems of the brain by its interference with acetylcholinesterase.
Most of the drugs used today to treat Alzheimer’s disease are agents that enhance the effects of the remaining ACh system. Nevertheless, it must be remembered that one certain characteristic of Alzheimer’s disease is a general reduction of aerobic metabolism in the brain. This results in a reduction in energy available for neuronal and muscular activity.
Because of the great affinity between fluorine and aluminum, it is possible that the greatest impairments of structure and function come about through the actions of charged and uncharged AlF complexes (AlFx). In the late 1970s and through the early 1990s there was considerable interest in the possibility that elemental aluminum was a major contributing factor to the development of dementia of the Alzheimer’s variety as well as to other neurological disorders. In a study of more than 3,500 French men and women above the age of 65 (Jacqmin et al. 1994), a significant decrease in cognitive abilities was found when their drinking water contained calcium, aluminum, and fluorine. Only aluminum showed any relation to cognitive impairment and that depended on the pH of the drinking water being below 7.3. Curiously, at higher pH values, a favorable effect on cognitive actions was found. In recent work with animals, aluminum-induced behavioral changes similar to those found in human dementia, as well as correlated histological changes in animals’ brains, were found (Miu et al. 2003). Active research continues at the cellular level on the neural mechanisms disturbed by aluminum (Becaria et al. 2003; Millan-Plano et al. 2003). On the epidemiological side there are inconsistencies in the results of different studies. For example, a recent review concludes that “the toxic effects of aluminum cannot be ruled out either, and thus exposure to aluminum should be monitored and limited as far as possible” (Suay and Ballester 2002). In addition to a depletion of acetylcholinesterase, fluoride produces alterations in phospholipid metabolism and/or reductions in the biological energy available for normal brain functions (see section later in this chapter on neurochemical effects). In addition, the possibility exists that chronic exposure to AlFx can produce aluminum inclusions with blood vessels as well as in their intima and adventitia. The aluminum deposits inside the vessels and those attached to the intima could cause turbulence in the blood flow and reduced transfer of glucose and O2 to the intercellular fluids. Finally histopathological changes similar to those traditionally associated with Alzheimer’s disease in people have been seen in rats chronically exposed to AlF (Varner et al. 1998).
ANIMAL STUDIES
Behavioral Changes
Studies of NaF
One of the most frequently cited and much discussed studies reporting a link between fluoride and behavior is by Mullenix et al. (1995). The study involved administering NaF to rats at different ages. Two groups of rats were exposed to NaF during gestation by subcutaneous injections given to pregnant dams. Other groups of rats received NaF in water beginning at weaning. Another set of rats was exposed to NaF in water in adulthood. Because of differences in the treatment regimes, procedures involved with the transport of animals at different ages, and other alterations in methods between the age groups, the data from the study are meaningful only if they are considered separately.
In “experiment 1,” pregnant dams were subcutaneously injected with NaF at 0.13 mg/kg either on gestational days 14-18 (one or two injections per day, for a total of nine injections) or on days 17-19 (three injections per day). In “experiment 2,” NaF at 75, 100, 125, or 175 mg/L was administered in the drinking water to rats at 21 days of age for 6-20 weeks. In “experiment 3,” 12-week-old rats were given NaF at 100 mg/L in drinking water for 5-6 weeks. Behavioral tests were performed on prenatally treated and weanling rats at 9 weeks of age, and adult-treated rats were tested at the end of their exposure period. Concentrations of fluoride in plasma in seven brain regions were measured at the time of sacrifice.
To appreciate the data generated by the testing procedures, some details of the testing methods and data analysis used in the Mullenix et al. study must be considered. The methods used were ones developed earlier to quantify animal behavior by using computer-based methods (Kernan et al. 1987, 1988: Kernan and Mullenix 1991). The basic procedures involved the following: The animals were tested in pairs consisting of a treated and a control rat. They were placed in a Plexiglas chamber divided in the middle by a Plexiglas wall to make two adjacent testing chambers. This wall had several holes in it. Thus, each rat could see, hear, and smell its pair-mate. The actual floor space available to each animal was approximately 10 in by 10 in. The chamber was an unusual trapezoidal design with the walls slanting outward from the floor. This shape was created to enhance the clarity of images of the rats recorded by two video cameras. One camera was placed above the testing chambers and another was off to one side. Both were aligned so as to encompass the testing areas of both animals. Sprague-Dawley albino rats were used in the experiments and, to further enhance the pictures, the side away from the horizontally placed camera was black. The floor was also black.
The two video cameras recorded the behavior of both animals simultaneously. The cameras were programmed to take still photos of the animals every second for the 15-minute testing period. Thus, the cameras sent 900 pictures of each animal during a single test period. The computer was programmed to detect five bodily positions, eight “modifiers” (apparently this term means an action with a presumptive goal), and several combinations of postures and modifiers. In all, the computer could record more than 100 combinations of positions, modifiers, and combinations of one or more of the measures indicating the “presumed intentions” of the animals (e.g., groom/attention). For each of these postures or actions or combinations, the number of times it was initiated, the total time spent doing it, and the distribution of the act throughout the 15-minute period were calculated separately for each rat.
In experiment 1, none of the rats treated on gestational days 14-18 showed any behavioral differences from controls. However, among rats treated on gestation days 17-19, male rats were reported to be more active than controls. The increase in activity was attributed to increased instances of grooming and head turning and not enhanced locomotor movement. Plasma concentrations of fluoride were comparable to those of the controls. Fluoride concentrations in the brain were not measured in this group.
In experiment 2, high mortality was observed in the highest treatment group (175 mg/L), and testing was discontinued at that concentration. Female rats exposed to NaF at 125 mg/L had fewer instances of sitting, spent less time sitting, had fewer head turns, and had fewer clusters of grooming bouts than controls. They also showed a reduction in the groom/attention composite index. Females exposed to fluoride in drinking water at 100 mg/ L for 6 weeks showed behavioral changes related to grooming, including reduced grooming bouts, reductions in persistent grooming periods, and the grooming/attention cluster. However, these effects were not seen among the females treated for longer periods (20 weeks). Among male rats, changes in behavior were observed only in the 125 mg/L group evaluated after 16 weeks of treatment. Changes included less sitting, less head turning, more standing, and reductions in grooming behavior. Standing and seeming attention postures were increased in these weanling-exposed rats. Measurements of fluoride in plasma showed an increase in concentration after 6 weeks of exposure to NaF at 100 mg/L in male and female rats. All seven areas of the brain analyzed showed increased concentrations of fluoride. As noted in Chapter 3, the accuracy of these measurements has been questioned (Whitford 1996), because other studies have shown that brain fluoride concentrations are considerably lower than, but proportionate to, those in plasma (Carlson et al. 1960; Whitford et al. 1979).
The computer program used in the behavior analyses also generated a statistic named “RS” that combines all the detected alterations in every recognized mode or modified mode of behavior. This overall index of change was reported as significant in females 6 weeks after the start of NaF treatment at concentrations of 100 and 125 mg/L. The statistic was not changed in males treated with NaF at a concentration of 125 mg/L for 11 weeks.
In experiment 3, only female rats showed behavioral changes compared with controls. Changes included reductions in sitting and grooming. Plasma fluoride concentrations were increased in males and females. Testing of fluoride concentrations in the brain found increased concentrations in the medulla of both sexes and in the hippocampal region of females. As noted above, the accuracy of these measurements has been questioned.
The results from these three experiments are difficult to interpret. One difficulty is interpreting the computer-derived categorization of activity patterns compared with behavioral descriptions commonly used by most animal researchers. For example, increased activity usually refers to increased locomotor activity measured in relatively large open fields or mazes. In the Mullenix et al. study, increased activity is characterized by head turning, grooming behaviors, and sniffing and exploration of the corners of the box, which traditionally are not characterized as part of locomotor activity. The small chambers in which the animals were tested would have prevented much locomotor movement at all.
Another aspect of the study that is a modifying issue is the stress-related experience of the rats before the experiments began. The transportation and associated handling of animals over long distances are known stressors to rats and mice. For experiment 1, the pregnant rats were shipped on day 6 of gestation and were housed singly thereafter. The rats used in experiment 2 were shipped to the laboratory at 17 days of age, along with their dams. The adult rats of experiment 3 were shipped at 10 weeks of age. Because the animals were from the Charles River Laboratories in Kingston, New York, the means of transportation to the laboratory in Boston was likely by truck. The transportation of animals by land or air has been shown to produce lasting effects on rodents (Isaacson et al. 2003). The histological effects of transportation and relocation include neuronal losses and substantial instances of shrunken or bloated cells, including some with condensed cytoplasmic inclusions. Other signs of stress and neural insult can be seen, including the presence of reactive microglia throughout the brain. These changes might well interact with later fluoride treatments. In essence, this means comparisons between groups can be legitimately made within the several experiments but not between them. Mullenix et al. (1995) interpreted their behavioral results to imply the interruption of hippocampal dysfunction. Another plausible interpretation is that the behavioral change might have involved alterations in the adrenal-pituitary axis (Gispen and Isaacson 1986).
The results of the Mullenix studies are difficult to compare with studies from other laboratories. The apparatus used has a unique configuration, the chambers were small, and the paired animals were in visual, olfactory, and auditory contact with each other. The data generated are largely derived in idiosyncratic ways by the hardware and software of a relatively complex computer program. From a practical standpoint, it would be extremely difficult for other investigators to replicate the study. The committee is aware there has been debate about the interpretation and significance of the findings of this study. For example, Ross and Daston (1995) note that decreased grooming can be an indication of illness. Because of the high concentrations of fluoride used in the study, it is possible that the animals had gastrointestinal or renal disturbances (Whitford and Taves 1973; Pashley et al. 1984; also see Chapter 9). As discussed above, the committee agrees there are difficulties with interpreting the results of the study, but those difficulties do not warrant dismissal of the results. The study provided some evidence that exposure to fluoride (prenatal, weaning, or in adulthood) might have affected the behavior of rats, albeit almost always in a gender-specific fashion.
In a different type of study, Swiss albino mice were treated with NaF at 30, 60, and 120-mg/L in water for 30 days and behavioral tests were performed daily 1 hour after treatment. The testing included akinesia, catalepsy, swim endurance, and simple maze tests. Animals in the 120 mg/L group scored more poorly in all the tests. Histological changes observed in the brains of these animals are discussed later in this chapter (Bhatnagar et al. 2002).
Paul et al. (1998) investigated the effects of NaF on the motor activity and coordination of female Wistar rats. The rats were treated with NaF at 20 or 40 mg/kg/day by gastric intubation for 2 months and were tested in an activity chamber and on a rota-rod apparatus. Only female rats were used because of the high mortality rates among males in preliminary studies. In both treatment groups, food intake and body weight gain were reduced in a dose-dependent manner. A reduction in spontaneous motor activity was based on results from an apparatus that recorded every type of movement, bodily adjustment, or twitch. This should not be confused with increased activity as measured by locomotor movements in a large arena. In the rota-rod motor coordination test, no significant changes were observed between the treated and control rats. There was a dose-related decrease in cholinesterase in the blood but not in the brain. Similar effects on motor activity have been observed in other studies in which rats were treated with NaF at 500 mg/L in drinking water. Alterations of acetylcholinesterase concentrations were found in the brain at this concentration (Ekambaram and Paul 2001, 2002).
Studies of AlF3
Varner et al. (1994) studied the effects of chronic administration of aluminum fluoride (AlF3), on the behavior of Long-Evans rats. AlF3was administered in drinking water at concentrations of 0.5, 5.0, or 50 mg/L. In terms of fluorine, these values translate into the equivalent of 0.34, 3.4, and 34 mg/L. The animals were between 130 and 154 days old at the beginning of the experiment and were maintained on this program for 45 weeks. In the animals treated with AlF3 at 5 and 50 mg/L, no differences in behavior were found in activity in an open field, in patterns of stride when walking, in spontaneous alternation of arms in a T-maze, in a motor coordination test, or in two tests of learning and memory in the Morris water maze. (Rats in the 0.5-mg/L group were too few to provide meaningful results.) The only behavioral change noted was a lack of preference of the location of a banana odor over the location of a lemon odor. Control animals generally prefer the banana odor. This overall lack of behavioral effects occurred in spite of extensive histological changes associated with neuronal damage and cell death in the hippocampus and other parts of the forebrain.
Anatomy
The complete analyses of the changes found in the brains of rats given one of the three doses of AlF3 used by Varner et al. (1994) were reported in a separate paper (Varner et al. 1993). All groups of the AlF3-exposed rats had significant losses of cells in the CA1 and CA3 areas of the hippocampus, but the losses were not dose dependent. Two types of cellular anomalies were found in the treated animals: (1) argentophilic cells throughout the hippocampus and dentate gyrus with considerable sparing of cells in the CA2 region; and (2) increased aluminum fluorescence in most of the brain, especially in the inner and outer linings of a large number of blood vessels, both large and small. Intravascular inclusions of aluminum particles were sometimes noted within blood vessels. Cells containing aluminum inclusions were not uncommon. This enhancement of aluminum deposits is not surprising because the amount of aluminum found in the brain was almost double that found in control animals.
Varner et al (1998) undertook a second study to determine the relative contribution of fluoride to the high mortality found in the 0.5-mg/L group of the earlier study, to extend the histological procedures used to evaluate the brains, and to determine whether the high death rates after this low dose would be found on replication. Three groups of nine adult rats were administered AlF3 at 0.5 mg/L, NaF at 2.1 mg/L (containing the same amount of fluoride as the AlF3 group), or double-distilled deionized water for 1 year. During that time six of nine animals drinking the AlF3 water died, three of the nine animals drinking the NaF died, and one animal from the control group died. Aluminum content in brain, kidney, and liver was measured by a direct current plasma technique modified for use with tissues containing substantial fat. Brains from both the NaF and the AlF3 groups had more than twice as much aluminum as the brains of the control animals. This supports the work of Strunecka et al. (2002) indicating that fluoride enhances the uptake of aluminum. But, the uptake was organ specific. There was no increase of aluminum found in the kidneys or liver. Sections from the brains of all animals were processed in a manner that allowed their staining with hematoxylin and eosin, the Morin stain for aluminum (and counterstained with cresyl violet), and a modified Bielschowsky silver stain as well as with antisera specific for IgM, ?-amyloid, or amyloid A.
There was a progressive decline in the appearance of the AlF3treated rats compared with the NaF or control animals before their demise. Their hair was sparse and their skin had a copper color. Toenails and teeth indicated a condition reflecting a hypermelanosis. Body weights, however, did not vary among the groups. Hemispheric differences in the brain were found in the distribution of aluminum using the Morin staining ultraviolet microscopic procedure. A greater amount of aluminum fluorescence was seen in layers 5 and 6 of the parietal neocortex and hippocampus of the left relative to the right hemisphere in the AlF3-treated rats. Areas CA3 and CA4 were the most affected regions of the hippocampus.
The occurrence of abnormal cells was also determined for all brains. Signs of neuronal anomalies included chromatin clumping, enhanced protein staining, pyknosis, vacuolation, ghost-like swollen appearances of cells, and enhanced silver staining in cell bodies and their processes. Both NaF and AlF3 treatments produced cellular distortions in cortical layers 2 and 3 of both hemispheres, but enhanced cellular abnormalities in layers 5 and 6 were found only in the left hemisphere. Both treatments also produced a diminished number of cells in the left CA3 region of the hippocampus but only the AlF3 treatment reduced cell numbers in this region of the left hemisphere. These observations are similar to previous findings reported in the brains of cats after intracerebroventricular administration of aluminum chloride (Crapper and Dalton 1973).
Both the AlF3 and the NaF treatments increased staining of neurons for IgM in the right hemisphere. No differences were found among the groups in the presence of IgM on the left side of the brain. Minor amounts of IgM were found in the hippocampus and dentate gyrus but without any group differences. The control group had few instances of ?-amyloid but the brains of the AlF3-treated animals demonstrated a bimodal distribution of deposits in the vasculature of the dorsal thalamus. Staining was either very high or nonexistent. The NaF-treated group showed a similar bimodality of accumulation of ?-amyloid in the right lateral posterior thalamic region.
The pattern of neuronal degeneration found by Varner et al. (1998) was also found in two other studies (Bhatnager et al. 2002; Shivarajashankara et al. 2002). In the study by Bhatnagar et al. (2002) described earlier in this chapter, the investigators observed a significant number of degenerated nerve cell bodies in hippocampal subregions CA3 and CA4 and in the dentate gyrus. Shivarajashankara et al. (2002) exposed Wistar rats to NaF in utero during the last week of gestation and for 10 weeks after birth. Animals received either 30 or 100 mg/L in their drinking water. At the end of the 10 weeks the animals were sacrificed and their brains were sectioned and stained with cresyl violet. Little change was seen in the 30-mg/L treated animals but the brains of the 100-mg/L treated animals showed large amounts of neurodegeneration. There were only a few normal appearing pyramidal cells in regions CA1 and CA3 of the hippocampus. Almost all the cells in these areas were pyknotic and showed intensely stained protein in their shrunken cytoplasm. Neuronal degeneration, but to a lesser degree, was found in the upper layers of neocortex, the amygdala, and the cerebellum. These areas were not extensively studied by Varner et al. (1998).
The interactions between fluoride and aluminum have been studied in laboratories and in the environment. There is evidence that fluoride enhances the uptake of aluminum and that aluminum reduces the uptake of fluoride (Spencer et al. 1980, Ahn et al. 1995). This complicates predicting the effect of exposure to aluminum- or fluorine-containing complexes in natural situations.
NEUROCHEMICAL EFFECTS AND MECHANISMS
A number of studies have examined biochemical changes in the brain associated with fluoride. For example, Guan et al. (1998) reported alterations in the phospholipid content of the brain of rats exposed to NaF at 30 or 100 mg/L for 3-7 months. The most prominent changes were found in phosphatidylethanolamine, phosphatidylcholine, and phosphatidylserine.
After 7 months of treatment, ubiquinone was clearly elevated, likely due as a compensatory reaction to the increase in free radicals in the brain. Fluoride has been shown to decrease the activities of superoxide dismutase (Guan et al. 1989) and glutathione peroxidase (Rice-Evans and Hoschstein 1981), the consequences being increased free radicals.
NaF injected subcutaneously into rabbits altered brain lipid metabolism (Shashi 1992b) and concentrations of protein, free amino acid, and RNA in the brain (Shashi et al. 1994).
Using slices of rat neocortex, Jope (1988) found that NaF stimulated the hydrolysis of phosphoinositide by activation of a G protein, Gp. This protein acts as a transducer between receptors and phospholipase C. He also found that a metal chelator added to the preparation eliminated this effect. This information and other observations led to the conclusion that the effective agent in the hydrolysis was an AlFxcomplex. Under his experimental conditions, the AlF4 was most likely formed from trace amounts of aluminum derived from the glass or from a fluorine-containing contaminant in a reagent. The addition of increasing amounts of aluminum did not increase the hydrolysis effect. In fact, adding substantial amounts of aluminum inhibited it. As in several types of experiment, it is the low aluminum fluoride concentrations that produce the greatest biochemical or physiological effects. In this regard, it is important to note that, even if aluminum bioavailability is low in rats and in other laboratory species, only a small amount is needed to produce untoward effects (Yokel et al. 2001).
Many of the untoward effects of fluoride are due to the formation of AlFx complexes. AlFx and BeFx complexes are small inorganic molecules that mimic the chemical structure of a phosphate. As such they influence the activity of phosphohydrolases and phospholipase D. Only micromolar concentrations of aluminum are needed to form AlFx(Sternweis and Gilman 1982). The G protein effects produced by AlFxare not limited to enzymes that bind phosphates or nucleoside-polyphosphate (Chabre 1990). AlFx also impairs the polymerization-depolarization cycle of tubulin. This could account for some of the intensely stained neurofilaments in cells in the brains of animals exposed to chronic NaF (Varner et al. 1993, 1998). AlFx appears to bind to enzyme-bound GDP or ADP, thus imitating GTP or ATP and, in a sense, generating “false messages” within the brain. This binding ability is probably due to the molecular similarities between AlF3(OH) and a phosphate group in the molecular structure, in particular, a tetrahedral arrangement (Strunecka and Patocka 2002).
G protein-coupled receptors mediate the release of many neural transmitters including the catecholamines, serotonin, ACh, and the excitatory amino acids. They also are involved in regulating glucagons, vasopressin, neuropeptides, endogenous opioids, prostaglandins, and other important systemic influences on brain and behavior. AlFx is also involved in regulating the pineal melatonin system as well as the thyroid-stimulating hormone-growth hormone connection. It has been said in this regard “every molecule of AlFx is the messenger of false information” (Strunecka and Patocka 2002, p. 275). This may be an accurate synopsis of the AlFxeffect at a single synapse, but the brain is a highly redundant and dispersed communication system containing millions of synapses. Because of this, observable alterations in mental or motor actions might require the formation of a multitude of false messages in a number of brain circuits acting over a prolonged period of time. Thus, the number of false messages required to disrupt an “action pattern” in the brain probably will vary according to the nature of the ongoing activities.
An especially important neurochemical transmitter that reaches almost all areas of the brain is ACh. As discussed above, some studies show that NaF and SiF inhibit cholinesterases, including acetylcholinesterase. The progressive accumulation of ACh at synaptic locations produced by the diminished esterase activity leads to a number of complex effects that can be summarized as an initial increase in stimulation of the target cells but ultimately leads to diminished stimulation—even a blockade of all activity. This earlier dialogue properly emphasized the behavioral importance of cholinergic activity in the brain and body more generally.
Long et al. (2002) reported changes in the number of acetylcholine receptors (nAChRs) in the rat brain due to fluoride. Rats were administered NaF in drinking water at 30 or 100-mg/L for 7 months. Decreased numbers of nAChR?7 subunits were found in the brains of rats from both treatment groups, but only the brains of the 100-mg/L group had diminished nAChR?4 subunits of this receptor. These results are of interest because changes in the nicotinic receptors have been related to the development of Alzheimer’s disease (Lindstrom 1997; Newhouse et al. 1997) and, in frontal brain areas, to schizophrenia (Guan et al. 1999).
FINDINGS
Human Cognitive Abilities
In assessing the potential health effects of fluoride at 2-4 mg/L, the committee found three studies of human populations exposed at those concentrations in drinking water that were useful for informing its assessment of potential neurologic effects. These studies were conducted in different areas of China, where fluoride concentrations ranged from 2.5 to 4 mg/L. Comparisons were made between the IQs of children from those populations with children exposed to lower concentration of fluoride ranging from 0.4 to 1 mg/L. The studies reported that while modal IQ scores were unchanged, the average IQ scores were lower in the more highly exposed children. This was due to fewer children in the high IQ range. While the studies lacked sufficient detail for the committee to fully assess their quality and their relevance to U.S. populations, the consistency of the collective results warrant additional research on the effects of fluoride on intelligence. Investigation of other mental and physiological alterations reported in the case study literature, including mental confusion and lethargy, should also be investigated.
Behavioral Effects on Animals
A few animal studies have reported alternations in the behavior of rodents after treatment with fluoride. However, the observed changes were not striking in magnitude and could have been due to alterations in hormonal or peptide activity. Animal studies to date have used conventional methodologies to measure learning and memory abilities or species-typical behaviors in novel locations. The tasks used to measure learning and memory did not require any significant mental effort. No studies were available on higher order mental functions, altered reactions to stress, responses to disease states, or supplemental reactions to known neurotoxins. Procedures are available that could test for cognitive functions, but they are labor intensive and have seldom been used in the past 60 years. One example is the reasoning test designed by Maier (1929), who found that even a small lesion of the neocortex impaired performance on the reasoning test (Maier 1932). A more recent example is the delayed matching to position test with different outcomes (Savage 2001), which have shown that damage to the hippocampus can affect learning.
Fluorosilicates
As noted in Chapter 2, exposure to fluorosilicates could occur under some conditions. There are reports that such chemicals enhance the uptake of lead into the body and brain, whereas NaF does not. Further research is needed to elucidate how fluorosilicates might have different biological effects from fluoride salts.
Neurochemical and Biochemical Changes
Lipids and phospholipids, phosphohydrolases and phospholipase D, and protein content have been shown to be reduced in the brains of laboratory animals subsequent to fluoride exposure. The greatest changes were found in phosphatidylethanolamine, phosphotidylcholine, and phosphotidylserine. Fluorides also inhibit the activity of cholinesterases, including acetylcholinesterase. Recently, the number of receptors for acetylcholine
has been found to be reduced in regions of the brain thought to be most important for mental stability and for adequate retrieval of memories.
It appears that many of fluoride’s effects, and those of the aluminofluoride complexes are mediated by activation of Gp, a protein of the G family. G proteins mediate the release of many of the best known transmitters of the central nervous system. Not only do fluorides affect transmitter concentrations and functions but also are involved in the regulation of glucagons, prostaglandins, and a number of central nervous system peptides, including vasopressin, endogenous opioids, and other hypothalamic peptides. The AlFx binds to GDP and ADP altering their ability to form the triphosphate molecule essential for providing energies to cells in the brain. Thus, AlFx not only provides false messages throughout the nervous system but, at the same time, diminishes the energy essential to brain function.
Fluorides also increase the production of free radicals in the brain through several different biological pathways. These changes have a bearing on the possibility that fluorides act to increase the risk of developing Alzheimer’s disease. Today, the disruption of aerobic metabolism in the brain, a reduction of effectiveness of acetylcholine as a transmitter, and an increase in free radicals are thought to be causative factors for this disease. More research is needed to clarify fluoride’s biochemical effects on the brain.
Anatomical Changes in the Brain
Studies of rats exposed to NaF or AlF3 have reported distortion in cells in the outer and inner layers of the neocortex. Neuronal deformations were also found in the hippocampus and to a smaller extent in the amygdala and the cerebellum. Aluminum was detected in neurons and glia, as well as in the lining and in the lumen of blood vessels in the brain and kidney. The substantial enhancement of reactive microglia, the presence of stained intracellular neurofilaments, and the presence of IgM observed in rodents are related to signs of dementia in humans. The magnitude of the changes was large and consistent among the studies. Given this, the committee concludes further research is warranted in this area, similar to that discussed at a February 2-3,1999, EPA workshop on aluminum complexes and neurotoxicity and that recommended for study by NTP (2002).
RECOMMENDATIONS
On the basis of information largely derived from histological, chemical, and molecular studies, it is apparent that fluorides have the ability to interfere with the functions of the brain and the body by direct and indirect means. To determine the possible adverse effects of fluoride, additional data from both the experimental and the clinical sciences are needed.
- The possibility has been raised by the studies conducted in China that fluoride can lower intellectual abilities. Thus, studies of populations exposed to different concentrations of fluoride in drinking water should include measurements of reasoning ability, problem solving, IQ, and short-and long-term memory. Care should be taken to ensure that proper testing methods are used, that all sources of exposure to fluoride are assessed, and that comparison populations have similar cultures and socioeconomic status.
- Studies of populations exposed to different concentrations of fluoride should be undertaken to evaluate neurochemical changes that may be associated with dementia. Consideration should be given to assessing effects from chronic exposure, effects that might be delayed or occur late-in-life, and individual susceptibility (see Chapters 2and 3 for discussion of subpopulations that might be more susceptible to the effects of fluoride from exposure and physiologic standpoints, respectively).
- Additional animal studies designed to evaluate reasoning are needed. These studies must be carefully designed to measure cognitive skills beyond rote learning or the acquisition of simple associations, and test environmentally relevant doses of fluoride.
- At the present time, questions about the effects of the many histological, biochemical, and molecular changes caused by fluorides cannot be related to specific alterations in behavior or to known diseases. Additional studies of the relationship of the changes in the brain as they affect the hormonal and neuropeptide status of the body are needed. Such relationships should be studied in greater detail and under different environmental conditions.
- Most of the studies dealing with neural and behavioral responses have tested NaF. It is important to determine whether other forms of fluoride (e.g., silicofluorides) produce the same effects in animal models.